BRP Peptide: A Natural Ozempic Alternative?
Stanford researchers used AI to discover BRP, a 12-amino-acid peptide that suppresses appetite and reduces fat in animals — with fewer side effects than Ozempic

Millions of people taking semaglutide — sold as Ozempic or Wegovy — know the trade-off: meaningful weight loss, but often at the cost of nausea, constipation, and significant muscle mass reduction. A new Stanford Medicine study published in Nature on March 5, 2025, introduces a naturally occurring molecule that may deliver comparable appetite suppression and fat loss — without those side effects. The molecule is called BRP, and it was found not by sifting through lab samples, but with the help of artificial intelligence.

What Is the BRP Peptide and Where Does It Come From?
BRP stands for BRINP2-related peptide, named after its parent protein, BPM/retinoic acid inducible neural specific 2 (BRINP2). It is a remarkably small molecule — just 12 amino acids long — that belongs to a class of signalling molecules called peptide hormones.
Peptide hormones originate from larger, biologically inactive proteins called prohormones. Prohormones are cleaved by enzymes into smaller, active peptide fragments; some of these fragments then act as hormones that regulate processes like appetite, metabolism, and blood sugar. BRP is one such fragment, produced when the enzyme prohormone convertase 1/3 cuts its parent protein at specific sites.
What makes BRP notable is its potency. When Stanford researchers screened 100 candidate peptides for their ability to activate lab-grown neuronal cells, BRP increased cellular activity tenfold over control cells. For comparison, GLP-1 — the peptide that semaglutide mimics — produced only a threefold increase. That signal was striking enough to push the team toward animal testing.
How AI Made the Discovery Possible
Traditional peptide discovery is slow and imprecise. Isolating peptide hormones from biological tissue is difficult because the target molecules are rare relative to the enormous number of protein fragments produced during normal cellular activity. Picking out the meaningful signals from that noise using conventional mass spectrometry techniques demands considerable time and resources.
The Stanford team built an algorithm called Peptide Predictor to cut through that complexity. Rather than working with physical tissue samples, Peptide Predictor scanned all 20,000 human protein-coding genes, looking for the specific amino acid sequences where prohormone convertase 1/3 is known to cleave. The algorithm then filtered for proteins that are secreted outside the cell — a hallmark of hormones — and that contain four or more potential cleavage sites.
That filter narrowed 20,000 genes down to 373 candidate prohormones, yielding 2,683 unique predicted peptides. From those, researchers selected 100 peptides with properties suggesting biological activity in the brain. The approach compressed what might have been years of bench work into a tractable screening exercise. "The algorithm was absolutely key to our findings," said senior author Katrin Svensson, PhD, assistant professor of pathology at Stanford Medicine.

Animal Study Results: Appetite, Fat Loss, and Fewer Side Effects
The results in animal models were pronounced. A single intramuscular injection of BRP, given to lean mice and minipigs before a meal, reduced food intake over the following hour by up to 50% in both species. Minipigs were included because their metabolism and eating patterns more closely resemble those of humans than mice do, making the cross-species consistency especially meaningful.
In the weight-loss phase of the study, obese mice received daily BRP injections for 14 days. Those animals lost an average of 3 grams — a loss attributable almost entirely to fat, not muscle — while untreated control animals gained roughly 3 grams over the same period. The treated mice also showed improved glucose and insulin tolerance, suggesting broader metabolic benefits beyond appetite control alone.
Perhaps equally important was what did not happen. Behavioural monitoring of mice and pigs found no significant differences in movement, water consumption, anxiety-like behaviour, or bowel activity between treated and untreated animals. There was no evidence of nausea or food aversion — side effects that are common with semaglutide and that lead many patients to reduce or discontinue their dose.
Why BRP Acts Differently to Semaglutide
The distinction lies in where and how each molecule works. Semaglutide mimics GLP-1, a peptide hormone whose receptor is distributed across multiple tissues — the brain, gut, pancreas, and more. That broad receptor distribution explains why semaglutide produces widespread effects: slowing gastric emptying, lowering blood sugar, and yes, causing nausea and digestive disruption.
BRP appears to act specifically in the hypothalamus, the brain region most directly responsible for regulating appetite and metabolism. "The receptors targeted by semaglutide are found in the brain but also in the gut, pancreas and other tissues," Svensson explained. "In contrast, BRP appears to act specifically in the hypothalamus, which controls appetite and metabolism."
Further physiological and brain activity studies confirmed that BRP activates metabolic and neuronal pathways that are separate from those activated by GLP-1 or semaglutide. This means BRP and semaglutide are not simply redundant — they engage the body's weight-regulation system through different mechanisms, which opens the possibility that the two could eventually be studied in combination, or that BRP could serve patients who cannot tolerate GLP-1-based therapies.
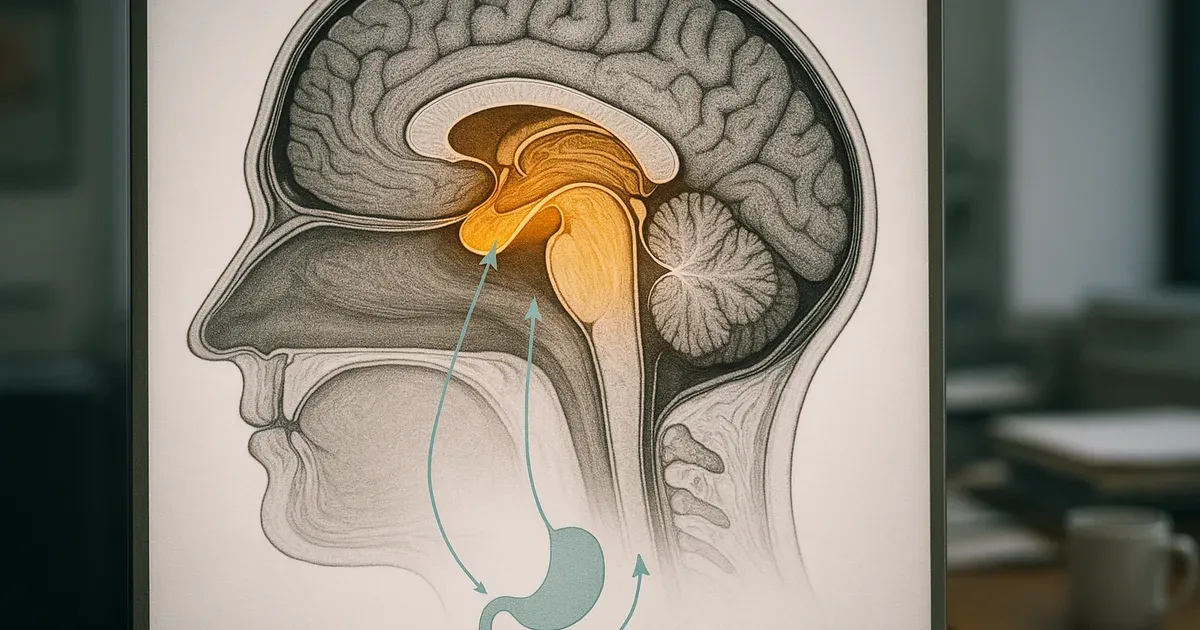
The Gut–Brain Axis: Why the Hypothalamus Matters
Appetite is not simply a matter of willpower. It is governed by a dense network of hormonal and neurological signals that flow between the digestive system, peripheral organs, and the brain — collectively described as the gut–brain axis. The hypothalamus sits at the centre of this network, integrating signals about energy balance and triggering the sensations of hunger and satiety.
Hormones and peptides that target the hypothalamus directly are therefore of particular interest in obesity research, because they interact with the system's master regulator without the off-target effects that come from activating receptors scattered throughout the body. BRP's apparent hypothalamic specificity positions it as a potentially cleaner pharmacological tool than existing GLP-1 receptor agonists.
Understanding exactly which cell-surface receptors bind BRP is the next step the Stanford team is pursuing. Identifying those receptors will clarify the precise pathway BRP uses, and may reveal further opportunities to develop or refine molecules that act through the same route.
What Comes Next: From Lab to Clinic
Svensson has co-founded a company, Merrifield Therapeutics, with the goal of launching clinical trials in humans. Both Svensson and lead author Laetitia Coassolo, PhD, hold patents on BRP peptides for the treatment of metabolic disorders.
One practical challenge the team is actively working on is extending how long BRP remains active in the body. Like most peptides, BRP is likely to be broken down relatively quickly after injection. Achieving a dosing schedule that is convenient for patients — weekly injections, for example, as is common with semaglutide — will probably require chemical modifications that protect the peptide from degradation without blunting its activity.
The research received funding from the National Institutes of Health, the SPARK Translational Research Program at Stanford, Stanford Bio-X, the American Heart Association, and several other bodies. Collaborating institutions included the University of California Berkeley, the University of Minnesota, and the University of British Columbia.

Frequently Asked Questions About BRP Peptide
Is BRP the same as Ozempic? No. BRP is a naturally occurring peptide identified in the human genome; semaglutide (Ozempic) is a synthetic drug that mimics GLP-1. Both suppress appetite and promote weight loss, but they act through different molecular pathways and different brain regions.
Has BRP been tested in humans yet? Not yet. All published results are from animal studies — mice and minipigs. Clinical trials in humans are being planned through Merrifield Therapeutics.
Why did BRP not cause nausea in animals? Researchers believe it is because BRP acts specifically in the hypothalamus rather than activating receptors in the gut and pancreas, which are associated with the gastrointestinal side effects of semaglutide. The hypothesis still needs to be confirmed in human trials.
Could BRP be used alongside semaglutide? The two molecules work through distinct pathways, which makes combination therapy a theoretical possibility. No combination studies have been published at this stage.
The Bottom Line
BRP peptide weight loss research represents one of the most promising developments in obesity pharmacology in recent years. Discovered through an AI-powered screening algorithm, this 12-amino-acid molecule matched — and in some measures exceeded — the appetite-suppressing effects of semaglutide in animal models, while apparently avoiding the nausea, gut disruption, and muscle loss that make GLP-1 drugs difficult for many patients to tolerate.
The mechanism is distinct and targeted: BRP appears to act directly on the hypothalamus, the brain's appetite-control hub, rather than broadcasting signals across the gut, pancreas, and multiple other tissues. That specificity could prove to be its defining advantage.
Human trials have not yet begun, and the history of obesity medicine is full of molecules that looked promising in rodents but failed to translate. Still, the cross-species consistency in mice and minipigs, the clean side-effect profile, and the mechanistic difference from existing drugs give BRP a genuinely compelling scientific rationale. The gut–brain research community will be watching Merrifield Therapeutics' clinical programme closely.
