Can Leaky Gut Cause Depression?
Yes — intestinal hyperpermeability allows LPS into systemic circulation, triggering neuroinflammation that suppresses hippocampal neurogenesis and disrupts serotonin metabolism. Plasma LPS levels are significantly elevated in major depressive disorder patients across multiple human studies.
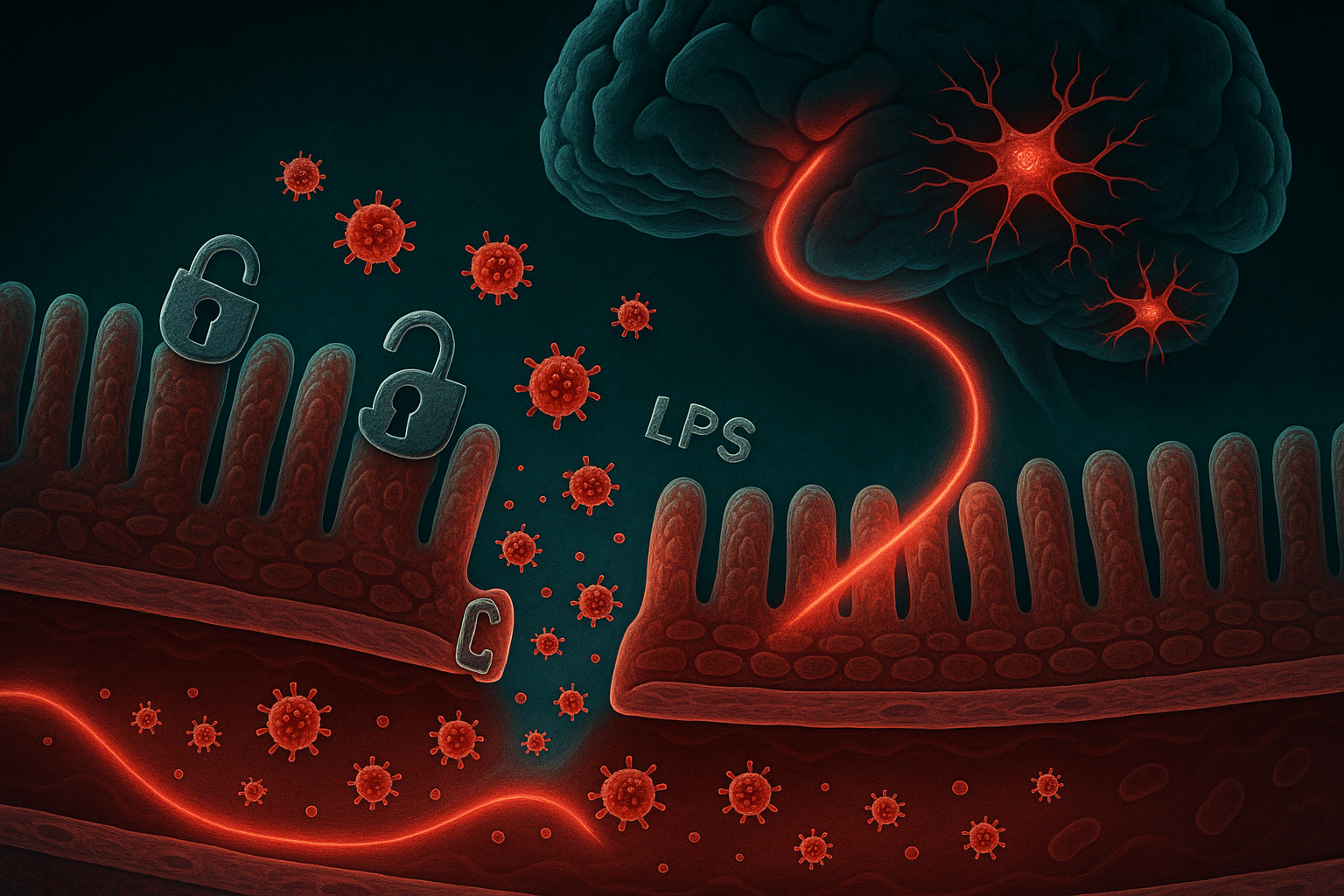
Yes — intestinal hyperpermeability ("leaky gut") is mechanistically linked to depression through the LPS-neuroinflammation pathway: increased gut barrier permeability allows bacterial lipopolysaccharides (LPS) to enter systemic circulation, triggering pro-inflammatory cytokines (IL-6, TNF-alpha, IL-1beta) that cross the blood-brain barrier, activate microglial cells, suppress hippocampal neurogenesis, and disrupt serotonin metabolism. Plasma LPS levels are significantly elevated in patients with major depressive disorder compared to healthy controls across multiple human studies.
Yes — intestinal hyperpermeability, commonly called "leaky gut," is linked to depression through a well-characterised biological pathway involving systemic inflammation and brain neurochemistry. This is not a fringe hypothesis: the LPS-neuroinflammation model of depression is now one of the leading mechanistic frameworks in biological psychiatry, supported by human biomarker data and interventional studies.
The Short Answer
When the gut barrier becomes permeable, bacterial lipopolysaccharides (LPS) — structural components of gram-negative bacteria cell walls — leak into the bloodstream. LPS triggers a systemic inflammatory response that crosses the blood-brain barrier, activates brain immune cells (microglia), suppresses new neuron growth in the hippocampus, and disrupts serotonin production — all core biological features of clinical depression. Multiple studies have found elevated circulating LPS and inflammatory markers in people with major depressive disorder.
The Science Behind It
The LPS pathway. The gut epithelium is protected by a single-cell-thick layer. When tight junction proteins (claudin, occludin, zonulin) are disrupted — by dysbiosis, antibiotic use, ultra-processed foods, or chronic stress — this barrier becomes permeable. Gram-negative bacteria (including E. coli and Bacteroides species) shed LPS as part of normal cell turnover. In a healthy gut, LPS is contained within the lumen. In a leaky gut, it passes into the portal vein and then systemic circulation.
Neuroinflammation. Circulating LPS binds to Toll-like receptor 4 (TLR4) on macrophages and triggers release of IL-6, TNF-alpha, IL-1beta, and IL-18 — pro-inflammatory cytokines that are known to cross the blood-brain barrier. These cytokines activate microglia, the brain's resident immune cells. Sustained microglial activation suppresses neurogenesis in the hippocampus — the brain region most implicated in depression — and shifts tryptophan metabolism away from serotonin synthesis toward the kynurenine pathway, producing neurotoxic quinolinic acid.
Human evidence. A 2022 meta-analysis in Translational Psychiatry found that plasma LPS-binding protein (LBP) — a marker of LPS exposure — was significantly elevated in patients with major depressive disorder compared to controls. A 2020 study in Brain, Behavior and Immunity found elevated intestinal fatty acid binding protein (I-FABP, a gut permeability marker) in depressed patients, correlating inversely with hippocampal volume on MRI.
What This Means For You
The leaky gut-depression link has practical implications: interventions that restore gut barrier integrity — reducing ultra-processed foods, increasing dietary fibre and fermented foods, addressing dysbiosis with targeted probiotics — may reduce the neuroinflammatory burden driving depressive symptoms. This is an emerging area with promising but not yet definitive clinical trial evidence specifically for depression treatment.
For the broader gut-brain axis context, see the Gut-Brain Axis Guide and the Gut Health & Mental Wellbeing hub. For anxiety-specific leaky gut effects, see Gut Health and Anxiety: What the Science Really Says.
Related Questions
What causes leaky gut in the first place? The most evidence-supported causes of increased intestinal permeability are: a high ultra-processed food diet (emulsifiers damage the mucus layer), antibiotic use (reduces protective microbial diversity), chronic psychological stress (activates stress hormones that impair tight junction proteins), excessive alcohol, and gut infections. Dysbiosis itself — reduced levels of Faecalibacterium prausnitzii, a key anti-inflammatory species — is both a cause and consequence of leaky gut.
Can you test for leaky gut? Yes — blood tests measuring I-FABP (intestinal fatty acid binding protein), zonulin, and LPS-binding protein are used in research settings to assess gut permeability. These are not widely available in routine clinical practice. Stool tests measuring calprotectin indicate gut inflammation but not specifically permeability.
Are leaky gut and IBS the same thing? No — intestinal hyperpermeability is a distinct physiological state that often overlaps with IBS but is not identical to it. Many people with elevated permeability have no digestive symptoms, yet still show systemic inflammatory and neurological effects.
