Best Foods for Gut Health and Mental Wellbeing: A Science-Backed Guide
The foods with the strongest evidence for gut-brain health: fermented foods (kefir, kimchi) increase microbial diversity and reduce neuroinflammation; prebiotic vegetables feed SCFA-producing bacteria; polyphenols act as selective prebiotics; omega-3s reduce neuroinflammation.…

Foods that support the gut-brain axis act through four primary mechanisms: providing prebiotic fibre that feeds SCFA-producing bacteria, delivering live fermented cultures that increase microbial diversity, supplying polyphenols that selectively promote beneficial *Lactobacillus* and *Bifidobacterium* populations, and providing omega-3 fatty acids that reduce neuroinflammation and support the blood-brain barrier. Dietary diversity — targeting 30+ different plant foods per week — is the single most evidence-supported intervention for improving microbiome composition and downstream mood outcomes.
The food on your plate does not just fuel your body — it feeds 38 trillion microbial organisms whose metabolic activity shapes your neurotransmitter levels, stress response, and emotional regulation. Dietary change is the most accessible, most studied, and — over appropriate timescales — most effective intervention for gut-brain axis health. But not all "healthy" foods are equally useful here. The evidence points to specific categories, specific mechanisms, and some clear hierarchies.
Fermented Foods: The Most Direct Microbiome Intervention
Fermented foods deliver live microorganisms directly to your gut — the most immediate way to influence microbial composition. The evidence for their mood and anxiety effects has strengthened considerably in recent years.
A landmark 2021 RCT published in Cell Host & Microbe by Wastyk et al. found that a high-fermented-food diet over 17 weeks increased microbiome diversity and reduced 19 inflammatory proteins — including IL-6 and IL-12p70 — more effectively than a high-fibre diet, despite the fibre diet producing larger microbiome compositional changes. This suggests fermented foods have direct anti-inflammatory effects beyond their microbiome impact.
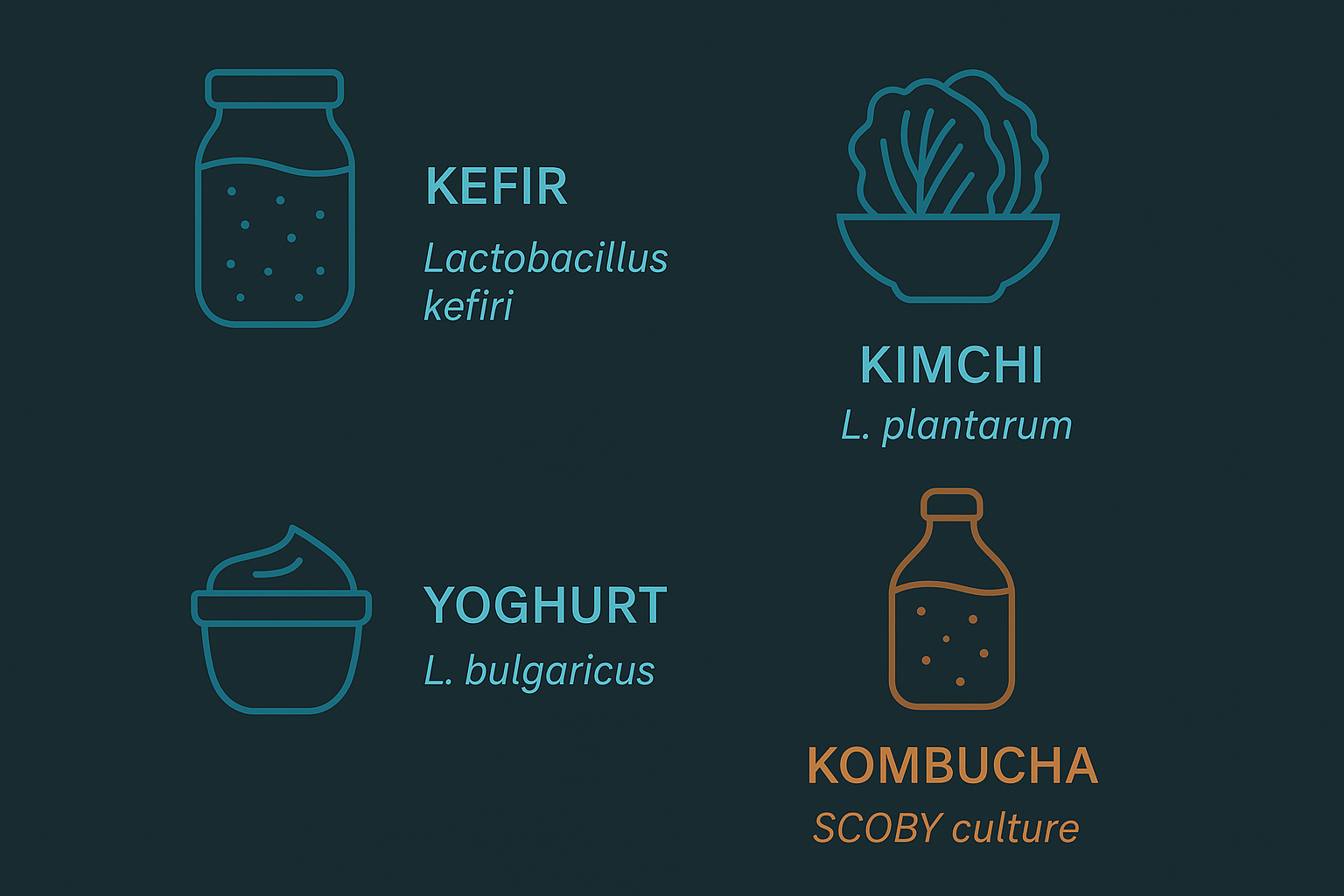
The foods with the strongest evidence base are:
Kefir — a fermented milk drink containing up to 61 different microbial species, including Lactobacillus kefiri, Lactococcus lactis, and Kluyveromyces marxianus. A 2022 RCT in Nutrients found 4 weeks of kefir consumption reduced anxiety and depression scores on validated scales compared to milk control.
Kimchi and sauerkraut — fermented vegetables rich in Lactobacillus plantarum and Leuconostoc mesenteroides. L. plantarum specifically produces GABA in measurable quantities and has been associated with stress reduction in human studies.
Plain live-culture yoghurt — the most accessible fermented food, but variable in bacterial count by brand. Look for products listing specific live strains rather than just "live cultures."
Kombucha and miso — moderate evidence, less studied than kefir and kimchi in clinical trials but with plausible mechanisms.
Prebiotic Foods: Feeding Your Existing Microbiome
Prebiotics are non-digestible dietary fibres that selectively feed beneficial gut bacteria, particularly Bifidobacterium, Faecalibacterium prausnitzii, and Roseburia intestinalis — species associated with SCFA production and reduced neuroinflammation.
A 2020 RCT in Psychopharmacology found that four weeks of prebiotic supplementation (fructooligosaccharides + galactooligosaccharides) reduced cortisol awakening response and attentional bias to negative stimuli — a cognitive marker of anxiety — compared to placebo in healthy volunteers.
The highest-prebiotic foods by inulin and FOS content are:
Jerusalem artichokes — the richest dietary source of inulin (14–19g per 100g raw). A small daily serving substantially increases Bifidobacterium counts within two weeks.
Garlic and onions — contain fructooligosaccharides and prebiotic inulin. Both promote Lactobacillus and Bifidobacterium growth and have documented anti-inflammatory effects via gut microbiome modulation.
Leeks, asparagus, and chicory root — consistent sources of inulin-type fructans. Chicory root is among the most concentrated sources (41g inulin per 100g dried).
Green bananas and cooked-then-cooled potatoes — high in resistant starch, which feeds Roseburia and Ruminococcus species — major butyrate producers.
Oats — rich in beta-glucan, a soluble fibre that increases Bifidobacterium and reduces inflammatory markers. A 2019 study in Gut found oat consumption significantly increased Akkermansia muciniphila, a keystone species associated with gut barrier integrity.
Polyphenol-Rich Foods: Prebiotic-Like Effects and Direct Anti-Inflammation
Polyphenols are plant secondary metabolites that act as selective prebiotics — they are metabolised by gut bacteria into bioactive compounds and simultaneously promote the growth of beneficial species while inhibiting pathogens.
Dark berries (blueberries, blackberries, raspberries) — among the richest dietary sources of anthocyanins. In vitro and human studies show anthocyanins increase Lactobacillus and Bifidobacterium populations and reduce Clostridium perfringens. A 2022 RCT in The American Journal of Clinical Nutrition found 6 weeks of blueberry consumption reduced inflammatory cytokines and improved mood scores.
Extra-virgin olive oil — contains oleocanthal and hydroxytyrosol, polyphenols that directly modulate Lactobacillus acidophilus and reduce LPS-driven neuroinflammation. The Mediterranean diet's robust evidence for mental health outcomes is substantially attributable to EVOO intake.
Dark chocolate (70%+ cocoa) — rich in flavonoids including epicatechin and catechin. A 2019 cross-sectional study in Depression & Anxiety involving 13,626 adults found that those consuming dark chocolate were 70% less likely to report depressive symptoms.
Green tea — contains L-theanine (an amino acid that increases alpha brain waves and promotes relaxation) and EGCG (a polyphenol that modulates the microbiome). A 2022 meta-analysis found daily green tea consumption was associated with lower anxiety scores.
Omega-3 Fatty Acids: Neuroinflammation and the Blood-Brain Barrier
Omega-3 fatty acids — primarily EPA (eicosapentaenoic acid) and DHA (docosahexaenoic acid) — reduce neuroinflammation by inhibiting the arachidonic acid pathway and support gut barrier integrity by reducing LPS translocation. They also increase Akkermansia muciniphila abundance, which strengthens the mucus layer protecting the intestinal epithelium.
A 2022 meta-analysis in Molecular Psychiatry covering 26 RCTs found that EPA supplementation (at least 1g/day) produced significant antidepressant effects, with the effect size comparable to that of antidepressant medication in populations with elevated baseline inflammatory markers.
Oily fish — salmon, mackerel, sardines, herring — are the richest dietary sources of EPA and DHA. Two to three servings per week is the level associated with mental health benefits in epidemiological studies.
Flaxseed and chia seeds — provide ALA (alpha-linolenic acid), which the body partially converts to EPA and DHA. Conversion efficiency is low (5–15%), making them a supplement to rather than replacement for oily fish.
For a practical meal framework built around these food categories, see The Gut-Brain Diet: A 7-Day Eating Plan for Mental Wellbeing. For information on how these dietary changes affect mood timelines, see How Long Does It Take to Improve Gut Health for Better Mood?
For the complete science behind why these foods work, see The Gut-Brain Axis: How Your Microbiome Controls Your Mood. For probiotic supplementation guidance that complements dietary changes, see What Are the Best Probiotics for Mental Health? For the complete topic hub, see the Gut Health & Mental Wellbeing topic page.
The 30-Plant Rule: Diversity Above All
The single most evidence-supported dietary recommendation for microbiome health is not any specific superfood — it is diversity. The American Gut Project, analysing microbiome data from over 11,000 participants, found that those consuming 30+ different plant foods per week had significantly greater microbiome diversity than those consuming 10 or fewer, regardless of whether they were vegan, vegetarian, or omnivorous.
This means variety matters more than volume: a small serving of eight different vegetables outperforms a large serving of the same two. Practically, counting different plant species — not servings — across the week (herbs, spices, legumes, grains, fruits, and vegetables all count) is the most actionable framework.

Frequently Asked Questions
Q: What foods improve gut health and mood? The foods with the strongest evidence for both gut health and mood are: fermented foods (kefir, kimchi, live-culture yoghurt), high-inulin prebiotic vegetables (Jerusalem artichokes, garlic, leeks), polyphenol-rich foods (dark berries, extra-virgin olive oil, dark chocolate), and omega-3 sources (oily fish). Dietary diversity — targeting 30+ different plant foods per week — is the most evidence-supported single strategy for microbiome improvement.
Q: Are fermented foods good for mental health? Yes — fermented foods increase gut microbial diversity, reduce systemic inflammation, and deliver GABA-producing bacteria (such as Lactobacillus plantarum) with direct mood-relevant effects. A 2021 RCT in Cell Host & Microbe found a high-fermented-food diet reduced 19 inflammatory proteins over 17 weeks, more effectively than a high-fibre diet alone.
Q: What is a psychobiotic? A psychobiotic is a live microorganism (probiotic) or prebiotic substrate that, when consumed in adequate amounts, produces a measurable mental health benefit through gut-brain axis mechanisms. The term was coined by Ted Dinan and John Cryan in 2013. Strains classified as psychobiotics include Lactobacillus rhamnosus JB-1, Bifidobacterium longum NCC3001, and the L. helveticus R0052 + B. longum R0175 combination — all with human trial evidence for anxiety or depression reduction.
Further Reading
For evidence-based dietary guidance from a leading academic nutrition institution, see The Nutrition Source at Harvard T.H. Chan School of Public Health, which covers dietary fibre, fermented foods, and gut health research. For a current review of the science on omega-3 fatty acids and health outcomes, see the NIH Office of Dietary Supplements omega-3 fact sheet.
Key Takeaways
- Fermented foods (kefir, kimchi, yoghurt) directly increase microbial diversity and reduce inflammation — the most immediate dietary intervention for the gut-brain axis
- Prebiotic-rich vegetables (Jerusalem artichokes, garlic, leeks) feed SCFA-producing bacteria linked to mood regulation and cortisol reduction
- Polyphenols from berries, olive oil, and dark chocolate act as selective prebiotics and have direct anti-inflammatory and mood-supporting effects
- Omega-3 fatty acids (EPA/DHA from oily fish) reduce neuroinflammation and support gut barrier integrity
- Dietary diversity — 30+ different plant foods per week — is the single most evidence-supported strategy for microbiome improvement
Foods that support the gut-brain axis act through four primary mechanisms: providing prebiotic fibre that feeds SCFA-producing bacteria, delivering live fermented cultures that increase microbial diversity, supplying polyphenols that selectively promote beneficial *Lactobacillus* and *Bifidobacterium* populations, and providing omega-3 fatty acids that reduce neuroinflammation and support the blood-brain barrier. Dietary diversity — targeting 30+ different plant foods per week — is the single most evidence-supported intervention for improving microbiome composition and downstream mood outcomes.
