Gut Health and Anxiety: What the Science Really Says
Gut dysbiosis is bidirectionally linked to anxiety via GABA depletion, glutamate excess, HPA axis dysregulation, and neuroinflammatory LPS cascades. Multiple RCTs confirm targeted probiotics reduce anxiety scores. Here's the complete evidence.
Gut dysbiosis — reduced microbial diversity and altered bacterial composition — is bidirectionally linked to generalised anxiety disorder via the gut-brain axis. Disrupted microbiomes produce excess glutamate and reduced GABA, impair vagal tone, and elevate circulating lipopolysaccharides that trigger neuroinflammatory cascades implicated in anxiety pathophysiology. Multiple randomised controlled trials demonstrate that targeted probiotic supplementation and dietary interventions reduce validated anxiety scores in human participants.
Anxiety disorders affect an estimated 301 million people globally, making them the most prevalent mental health condition on Earth. Yet for most of that history, treatment has focused almost exclusively on the brain — with SSRIs, CBT, and anxiolytic medications. A growing body of research now points to a different organ as a critical but overlooked player: the gut. The relationship between your microbiome and your anxiety is not one-way, and it is not metaphorical. It is physiological, measurable, and — crucially — modifiable.
How Gut Dysbiosis Drives Anxiety
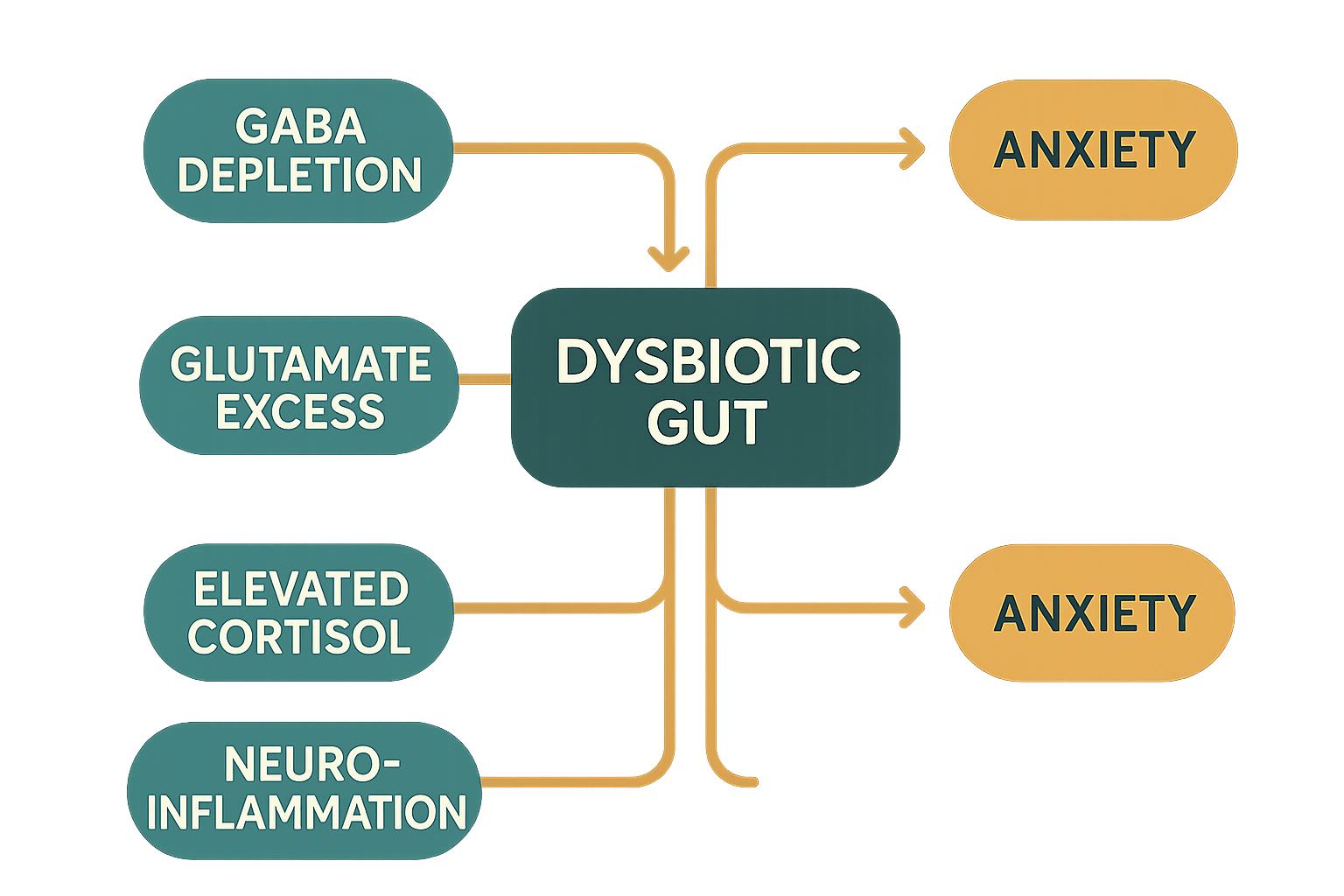
Gut dysbiosis describes a state of microbial imbalance — reduced diversity, depletion of beneficial species, or overgrowth of pathogenic or inflammatory bacteria. It drives anxiety through several convergent mechanisms.
GABA disruption. Lactobacillus rhamnosus and other Lactobacillus species produce GABA — the brain's primary inhibitory neurotransmitter — directly in the gut, and signal its production in the brain via vagal pathways. A landmark 2011 study in PNAS found that mice fed L. rhamnosus JB-1 showed dramatically reduced anxiety behaviour and altered GABA receptor expression in the brain compared to controls. When the vagus nerve was severed, these effects disappeared — confirming the gut-vagus-brain pathway.
Glutamate excess. In dysbiotic states, overgrowth of certain proteobacteria increases glutamate (the brain's primary excitatory neurotransmitter) production. Elevated glutamate signalling in the amygdala — the brain's threat-detection centre — is a core feature of anxiety neurobiology.
Elevated cortisol reactivity. As detailed in our gut-brain axis guide, dysbiosis impairs HPA axis calibration, resulting in exaggerated cortisol responses to mild stressors — a physiological signature of anxiety disorders.
Tryptophan diversion. Gut bacteria heavily influence how tryptophan is metabolised. In dysbiotic states, tryptophan is preferentially shunted down the kynurenine pathway rather than towards serotonin synthesis. Kynurenine metabolites — particularly quinolinic acid — are neurotoxic and have been found elevated in cerebrospinal fluid of patients with anxiety disorders (study: Frontiers in Psychiatry, 2022).
How Anxiety Disrupts Your Gut
The relationship runs in both directions. Anxiety does not just follow from gut problems — it actively worsens them, creating a reinforcing cycle that can be difficult to break without addressing both sides simultaneously.
Acute psychological stress activates the sympathetic nervous system ("fight or flight"), which immediately reduces blood flow to the digestive tract, alters gut motility (causing either constipation or diarrhoea), and increases gut permeability within hours. A 2020 study in Gut found that just 30 minutes of an acute stress protocol measurably increased intestinal permeability markers in healthy volunteers.
Chronic anxiety maintains elevated glucocorticoid levels that suppress the growth of beneficial Bifidobacterium and Lactobacillus species — exactly the bacteria most associated with GABA production and mood regulation. A 2021 study in Brain, Behavior and Immunity found that individuals with generalised anxiety disorder showed significantly lower Lactobacillus and Bifidobacterium counts and higher Enterobacteriaceae counts compared to non-anxious controls, even after controlling for diet and lifestyle.
This bidirectionality is clinically important: treating anxiety through stress reduction alone may not be sufficient if the gut microbiome has been sufficiently disrupted. Conversely, gut-targeted interventions may support — but are unlikely to replace — psychological approaches to anxiety treatment.
The Human Evidence: Probiotics and Anxiety
The strongest clinical evidence for a gut-anxiety link comes from randomised controlled trials using probiotic interventions.
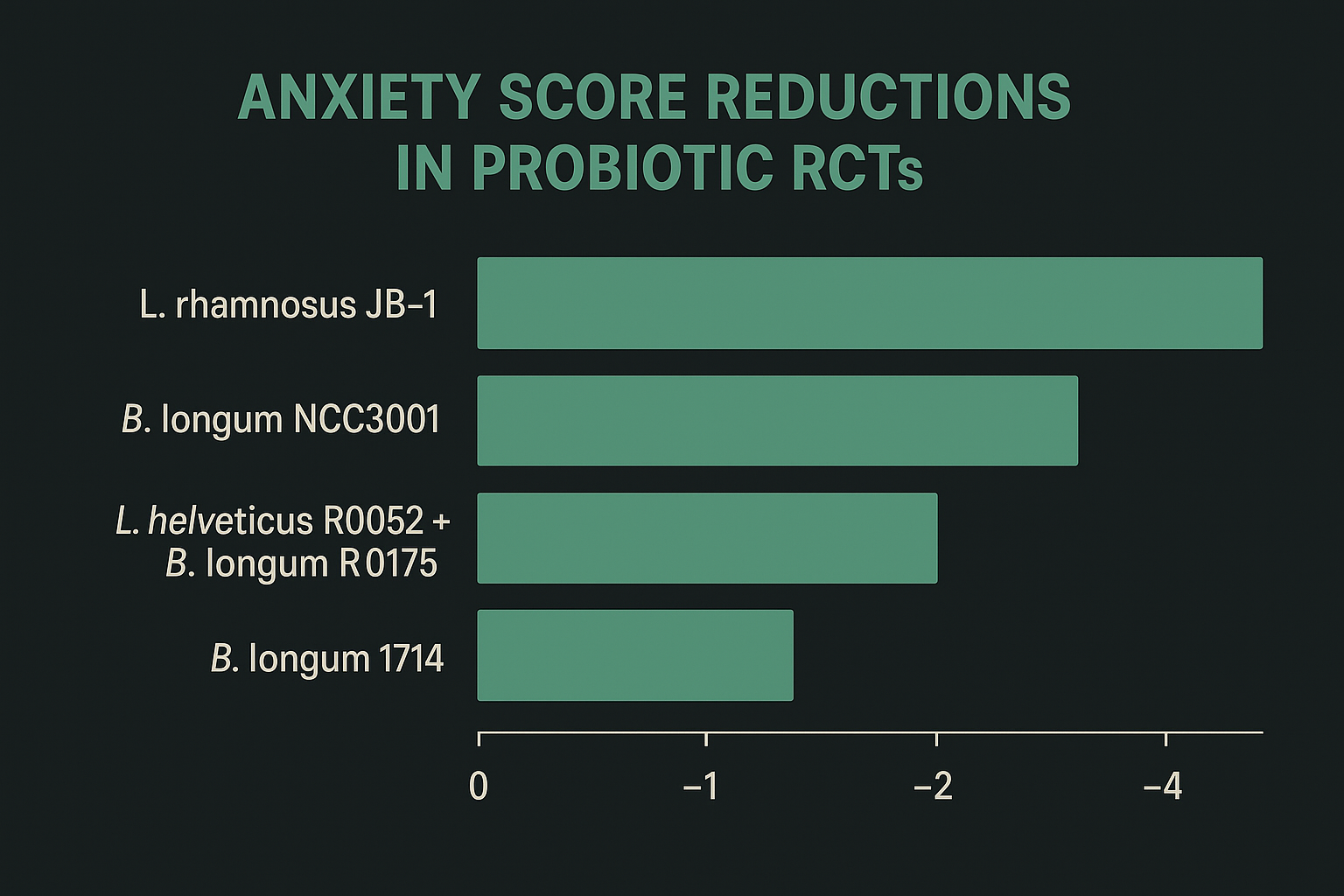
A 2019 meta-analysis in BMJ Nutrition covering 15 RCTs found that probiotic supplementation produced significant reductions in anxiety symptoms compared to placebo, with the greatest effects seen in studies using multi-strain products and durations of eight weeks or more. The most studied strains showing anxiolytic effects are Lactobacillus rhamnosus JB-1, Bifidobacterium longum NCC3001, and Lactobacillus helveticus R0052 combined with Bifidobacterium longum R0175.
A 2022 RCT published in Nutrients found that eight weeks of B. longum 1714 supplementation in healthy volunteers under academic exam stress significantly reduced cortisol awakening response, subjective stress, and anxiety scores on the Hamilton Anxiety Scale compared to placebo.
The evidence is promising but not yet conclusive for clinical anxiety disorders — most studies use subclinical or healthy-stressed populations. Current research positions probiotics as a credible adjunct to established anxiety treatments, not a standalone replacement.
For specific strain recommendations, see What Are the Best Probiotics for Mental Health? For the complete topic hub, see the Gut Health & Mental Wellbeing topic page.
Dietary Patterns and Anxiety: What the Evidence Shows
Beyond probiotics, whole dietary patterns show robust associations with anxiety risk.
The Mediterranean diet — rich in vegetables, legumes, olive oil, fish, and fermented dairy — has been associated with a 33% lower risk of anxiety disorders in a 2020 systematic review of 20 prospective studies (Nutritional Neuroscience). The high fibre content feeds SCFA-producing bacteria, while polyphenols from olive oil and red wine (resveratrol, hydroxytyrosol) directly modulate Lactobacillus and Bifidobacterium growth.
Ultra-processed food diets show the reverse pattern: a 2023 study in The British Journal of Psychiatry involving 10,359 participants found that each 10% increase in ultra-processed food as a proportion of diet was associated with a 12% higher odds of clinically significant anxiety symptoms.
Fermented foods specifically have emerging evidence: a 2021 RCT in Cell Host & Microbe found that a high-fermented-food diet (kefir, kimchi, kombucha, fermented vegetables) over 17 weeks increased microbiome diversity and reduced inflammatory markers compared to a high-fibre diet alone.
For a practical food-by-food breakdown, see Best Foods for Gut Health and Mental Wellbeing. For specific leaky gut-depression connections, see Can Leaky Gut Cause Depression?
Frequently Asked Questions
Q: Can gut bacteria cause anxiety? Yes — gut bacterial imbalances (dysbiosis) can contribute to anxiety through multiple physiological mechanisms: reduced GABA production by Lactobacillus species, elevated glutamate from proteobacteria overgrowth, impaired HPA axis calibration, and neuroinflammatory cascades driven by increased gut permeability. Multiple animal and human studies support this link, though dysbiosis is one of many factors in anxiety pathophysiology.
Q: What probiotics help with anxiety? The strains with the strongest human trial evidence for anxiety reduction are Lactobacillus rhamnosus JB-1, Bifidobacterium longum NCC3001, and the combination of Lactobacillus helveticus R0052 with Bifidobacterium longum R0175. Clinical trials typically use 8–12 week supplementation periods. Multi-strain products generally show larger effects than single-strain products in meta-analyses.
Q: Does anxiety damage gut health? Yes — anxiety activates the sympathetic nervous system and elevates cortisol, which impairs gut barrier integrity, reduces blood flow to the gut, suppresses beneficial Lactobacillus and Bifidobacterium populations, and increases intestinal permeability. This creates a reinforcing cycle: anxiety worsens dysbiosis, and dysbiosis amplifies anxiety signals via the gut-brain axis.
Further Reading
For clinical information on anxiety disorders and current treatment approaches, see the Anxiety and Depression Association of America (ADAA). For an overview of current evidence on probiotic supplementation, see the NIH National Center for Complementary and Integrative Health probiotics fact sheet.
Key Takeaways
- Gut dysbiosis drives anxiety via GABA depletion, glutamate excess, elevated cortisol reactivity, and tryptophan diversion — all measurable, physiological mechanisms
- Anxiety simultaneously damages gut health by increasing permeability and suppressing beneficial bacteria, creating a self-reinforcing cycle
- Multiple RCTs confirm probiotic supplementation (especially L. rhamnosus, B. longum) reduces anxiety scores — strongest evidence with 8+ week protocols
- Mediterranean dietary patterns are associated with significantly lower anxiety risk in large prospective studies
- Gut-targeted interventions are best understood as adjuncts to, not replacements for, established anxiety treatments
Gut dysbiosis — reduced microbial diversity and altered bacterial composition — is bidirectionally linked to generalised anxiety disorder via the gut-brain axis. Disrupted microbiomes produce excess glutamate and reduced GABA, impair vagal tone, and elevate circulating lipopolysaccharides that trigger neuroinflammatory cascades implicated in anxiety pathophysiology. Multiple randomised controlled trials demonstrate that targeted probiotic supplementation and dietary interventions reduce validated anxiety scores in human participants.
