The Gut-Brain Axis: How Your Microbiome Controls Your Mood
The gut-brain axis is a bidirectional communication network linking your microbiome to your brain via the vagus nerve, HPA axis, and immune pathways. Gut bacteria produce ~90% of serotonin and directly influence GABA, dopamine, and cortisol — making microbial diversity a direct determinant of mood.
The gut-brain axis is a bidirectional communication network between the intestinal microbiome and the central nervous system, operating via the vagus nerve, enteric nervous system, hypothalamic-pituitary-adrenal (HPA) axis, and immune signalling pathways. Gut bacteria synthesise approximately 90% of the body's serotonin and influence dopamine, GABA, and cortisol regulation, making microbial diversity a direct determinant of mood, stress resilience, and cognitive function.
Your gut contains more neurons than your spinal cord. The enteric nervous system — sometimes called the "second brain" — lines the entire digestive tract with over 500 million nerve cells, forming the most complex neural network outside the skull. But the gut is not simply a parallel brain: it communicates continuously with the organ above it, and the nature of that conversation determines far more about your mental state than most people realise.
What Is the Gut-Brain Axis?
The gut-brain axis is the continuous two-way communication system linking your gastrointestinal tract and your brain. It is not a single pathway but a multi-channel network operating simultaneously through neural, hormonal, immune, and metabolic signals.
The four primary channels are: the vagus nerve (direct neural highway); the enteric nervous system (the gut's autonomous nerve network); the hypothalamic-pituitary-adrenal (HPA) axis (the stress response system); and the immune system, through which gut bacteria modulate neuroinflammation. Each channel transmits information in both directions — the gut influences the brain, and the brain influences the gut — creating a feedback loop that connects digestive health to emotional regulation, cognition, and stress resilience.
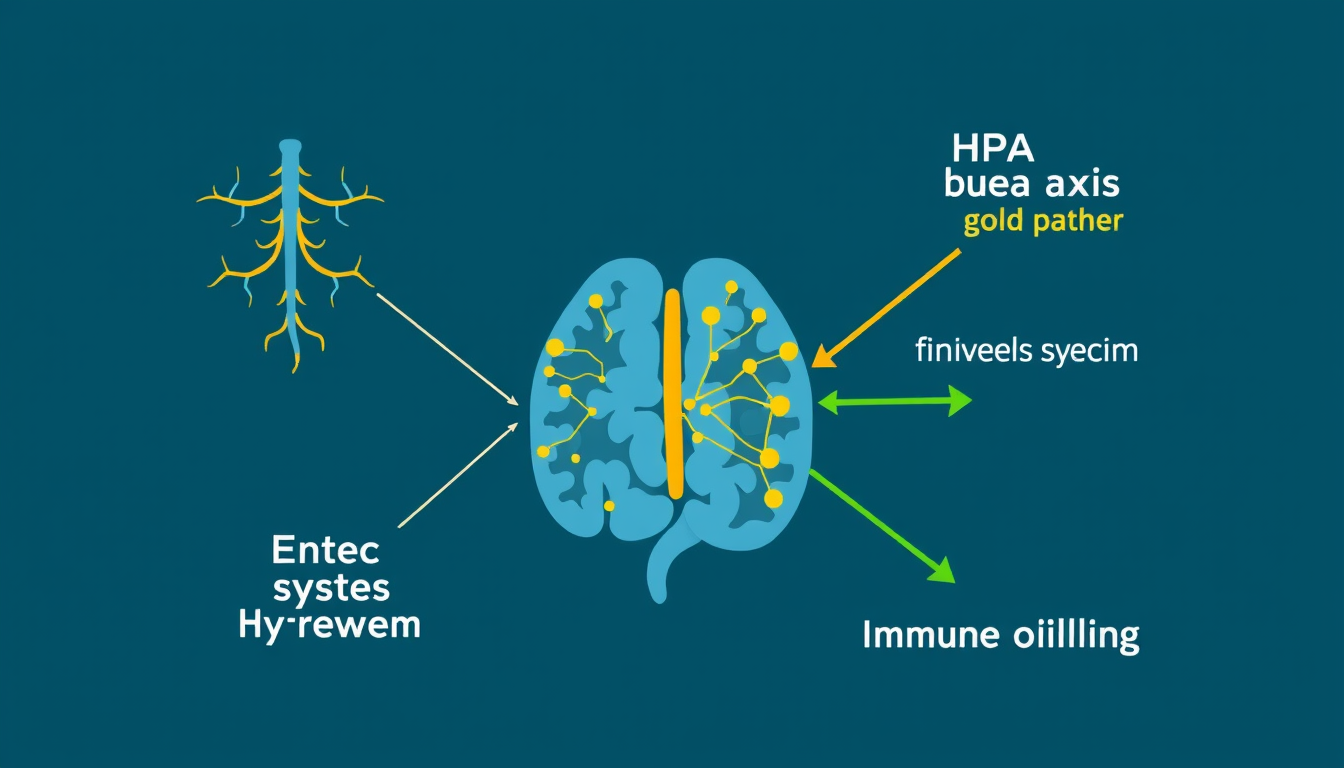
The Vagus Nerve: The Main Highway
The vagus nerve is the longest cranial nerve in the human body, running from the brainstem through the chest and abdomen to innervate the heart, lungs, and entire gastrointestinal tract. It is the primary anatomical highway of the gut-brain axis.
Approximately 80–90% of vagal fibres carry signals upward — from the gut to the brain — not the other way around. This asymmetry is significant: it means the gut is, in neurological terms, primarily a sender rather than a receiver. The vagus nerve continuously monitors gut wall tension, mucosal chemistry, and microbial metabolites, transmitting that information to the brainstem nucleus tractus solitarius, and from there to the limbic system, prefrontal cortex, and hypothalamus — structures governing emotion, decision-making, and the stress response.
A 2021 study in Cell demonstrated that specific gut bacteria activate vagal afferent neurons directly through epithelial neuropods — specialised sensory cells in the gut lining that synapse with vagal nerve endings within milliseconds, faster than any hormonal signal. The identity and abundance of the bacteria present therefore directly shapes what the vagus reports to the brain.
Serotonin, Dopamine, and the Gut's Neurochemical Factory
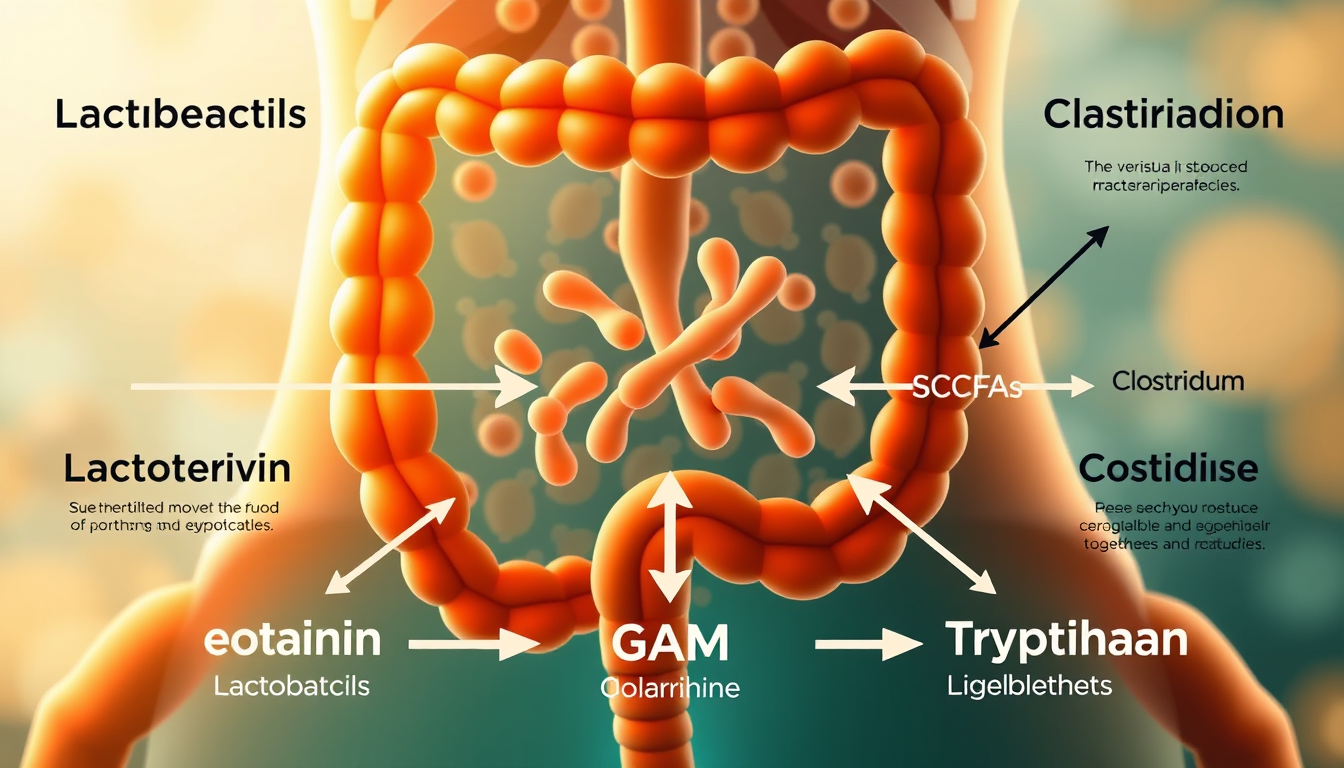
The gut produces approximately 90% of the body's serotonin, synthesised by enterochromaffin cells in the intestinal epithelium under instruction from gut bacteria — primarily Clostridium species and Enterococcus faecalis. This serotonin does not cross the blood-brain barrier, but it regulates gut motility, influences vagal signalling, and modulates platelet function in ways that affect systemic mood chemistry.
Gut bacteria also produce or stimulate production of: GABA (gamma-aminobutyric acid), the primary inhibitory neurotransmitter in the brain, linked to anxiety reduction; dopamine precursors including L-DOPA, with Bacillus species identified as producers; short-chain fatty acids (SCFAs) including butyrate, propionate, and acetate, which cross the blood-brain barrier and exert direct neuroprotective effects; and tryptophan metabolites along the kynurenine pathway, which influence neuroinflammation and depressive symptomatology.
A 2019 study in Nature Microbiology analysing 1,054 individuals found that depletion of Coprococcus and Dialister bacteria was consistently associated with lower quality of life scores and depressive symptoms, independent of antidepressant use — providing direct human evidence that microbiome composition influences mood neurochemistry.
The HPA Axis and Gut-Driven Stress Response
The hypothalamic-pituitary-adrenal (HPA) axis governs the body's cortisol stress response. Gut bacteria regulate HPA axis sensitivity in ways that determine how intensely you respond to psychological stress.
Germ-free animal studies have been pivotal here: mice raised without any gut microbiome exhibit exaggerated HPA responses to mild stressors — releasing two to three times the cortisol of conventionally colonised mice. When these germ-free mice are given a microbiome transplant from normal mice early in life, HPA sensitivity normalises. This demonstrates that microbial colonisation in early life calibrates the stress response system — and that dysbiosis can leave that system permanently over-sensitised.
In humans, a 2020 study in Psychoneuroendocrinology found that individuals with lower microbial diversity showed higher morning cortisol awakening responses and greater self-reported stress reactivity, controlling for lifestyle variables. The mechanisms involve microbial regulation of glucocorticoid receptor expression in the hippocampus, a process mediated by SCFAs and microbial metabolites acting on epigenetic pathways.
Neuroinflammation: The Leaky Gut Pathway
When the gut barrier becomes permeable — a condition known as intestinal hyperpermeability or "leaky gut" — bacterial lipopolysaccharides (LPS) from gram-negative bacteria enter systemic circulation. LPS triggers the release of pro-inflammatory cytokines including IL-6, TNF-alpha, and IL-1beta, which cross the blood-brain barrier and activate microglial cells — the brain's resident immune cells.
Sustained microglial activation suppresses neurogenesis in the hippocampus (the brain region most associated with depression), disrupts serotonin metabolism, and increases glutamate excitotoxicity. This neuroinflammatory cascade is now considered one of the primary biological mechanisms linking gut dysbiosis to depression. A 2022 meta-analysis in Translational Psychiatry found that plasma LPS levels were significantly elevated in patients with major depressive disorder compared to healthy controls.
For more on this specific pathway, see Can Leaky Gut Cause Depression?
What Disrupts the Gut-Brain Axis?
Several factors impair gut-brain axis function by reducing microbial diversity or damaging gut barrier integrity:
Antibiotics — broad-spectrum antibiotics can reduce gut microbial diversity by up to 30% and alter the gut-brain axis for months after a course ends, with some effects persisting for years.
Ultra-processed foods — diets high in emulsifiers (polysorbate 80, carboxymethylcellulose) directly damage the mucus layer protecting the gut epithelium, increasing LPS translocation.
Chronic stress — activates the sympathetic nervous system, altering gut motility and reducing populations of beneficial Lactobacillus and Bifidobacterium species.
Sleep disruption — the gut microbiome has its own circadian rhythm. Chronic sleep disruption desynchronises microbial activity, reducing SCFA production and increasing inflammatory markers.
Low dietary fibre — gut bacteria ferment fibre to produce SCFAs. Diets providing less than 25g of fibre daily starve the microbiome of its primary fuel, reducing Faecalibacterium prausnitzii — the single most anti-inflammatory bacterial species identified in human studies.
Practical Implications: Can You Improve Your Mood by Improving Your Gut?
The short answer is yes — with appropriate caveats about individual variation and timescales.
A 2022 randomised controlled trial published in Nature Communications found that a high-fibre, fermented-food diet increased microbiome diversity and reduced inflammatory markers over 17 weeks compared to a standard diet, with participants reporting significant improvements in mood and stress resilience. A 2023 meta-analysis in JAMA Psychiatry covering 34 RCTs found probiotic supplementation produced statistically significant reductions in both depression and anxiety scores.
Practical interventions with the strongest evidence base include: increasing dietary fibre diversity (targeting 30+ different plant foods per week); regular consumption of fermented foods (kefir, kimchi, yoghurt with live cultures); reducing ultra-processed food intake; and addressing sleep quality. For specific dietary guidance, see Best Foods for Gut Health and Mental Wellbeing. For targeted probiotic choices, see What Are the Best Probiotics for Mental Health?
For the full picture on how gut health specifically intersects with anxiety, see Gut Health and Anxiety: What the Science Really Says. For the complete topic hub covering all gut-brain research in one place, see the Gut Health & Mental Wellbeing topic page.
Frequently Asked Questions
Q: How does the gut communicate with the brain? The gut communicates with the brain through four simultaneous channels: the vagus nerve (direct neural signalling), the enteric nervous system, the HPA stress axis (via cortisol and glucocorticoids), and immune pathways (via cytokines and bacterial metabolites crossing the blood-brain barrier). The vagus nerve alone carries approximately 500 million signals per day between gut and brain.
Q: How much serotonin is made in the gut? Approximately 90–95% of the body's total serotonin is synthesised in the gut, produced by enterochromaffin cells in the intestinal epithelium under direct influence from gut bacteria — particularly Clostridium species. This serotonin does not cross the blood-brain barrier but regulates gut motility, influences vagal signalling, and affects peripheral mood-related processes.
Q: Can you improve your mood by fixing your gut? Yes — clinical evidence supports this. A 2023 meta-analysis in JAMA Psychiatry covering 34 randomised controlled trials found probiotic supplementation produced statistically significant reductions in depression and anxiety scores. Dietary interventions increasing fibre diversity and fermented food intake have also demonstrated measurable mood improvements in RCTs, typically over 4–17 weeks.
Further Reading
For a clinical overview of gut-brain axis research and its implications for psychiatric care, see the National Institute of Mental Health (NIMH) — Depression overview and the Johns Hopkins Medicine — The Gut-Brain Connection, a patient-facing overview from one of the world's leading academic medical centres.
Key Takeaways
- The gut-brain axis is a multi-channel bidirectional communication network — not a single pathway
- 80–90% of vagal signals travel upward from gut to brain, making the gut primarily a sender of information
- Gut bacteria produce ~90% of serotonin and directly influence GABA, dopamine precursors, and cortisol regulation
- Dysbiosis and gut barrier disruption drive neuroinflammation via the LPS-cytokine pathway — a key mechanism in depression
- Diet, probiotics, and sleep all have clinical evidence for improving gut-brain axis function and mood outcomes
The gut-brain axis is a bidirectional communication network linking intestinal microbiota to the central nervous system via the vagus nerve, enteric nervous system, HPA axis, and immune pathways. Gut bacteria produce approximately 90% of the body's serotonin and influence dopamine, GABA, and cortisol regulation, making microbial diversity a direct determinant of mood, stress resilience, and cognitive function.
