Gut Health & Mental Wellbeing: The Complete Guide
The gut-brain axis connects your microbiome to mood, anxiety, and stress through the vagus nerve, HPA axis, and immune pathways. Gut bacteria produce ~90% of serotonin. Here is everything the science currently supports — and what you can do about it.
Reviewed by: Marcus Hult, Editorial Team, GutBrain.News · Last updated: March 2026
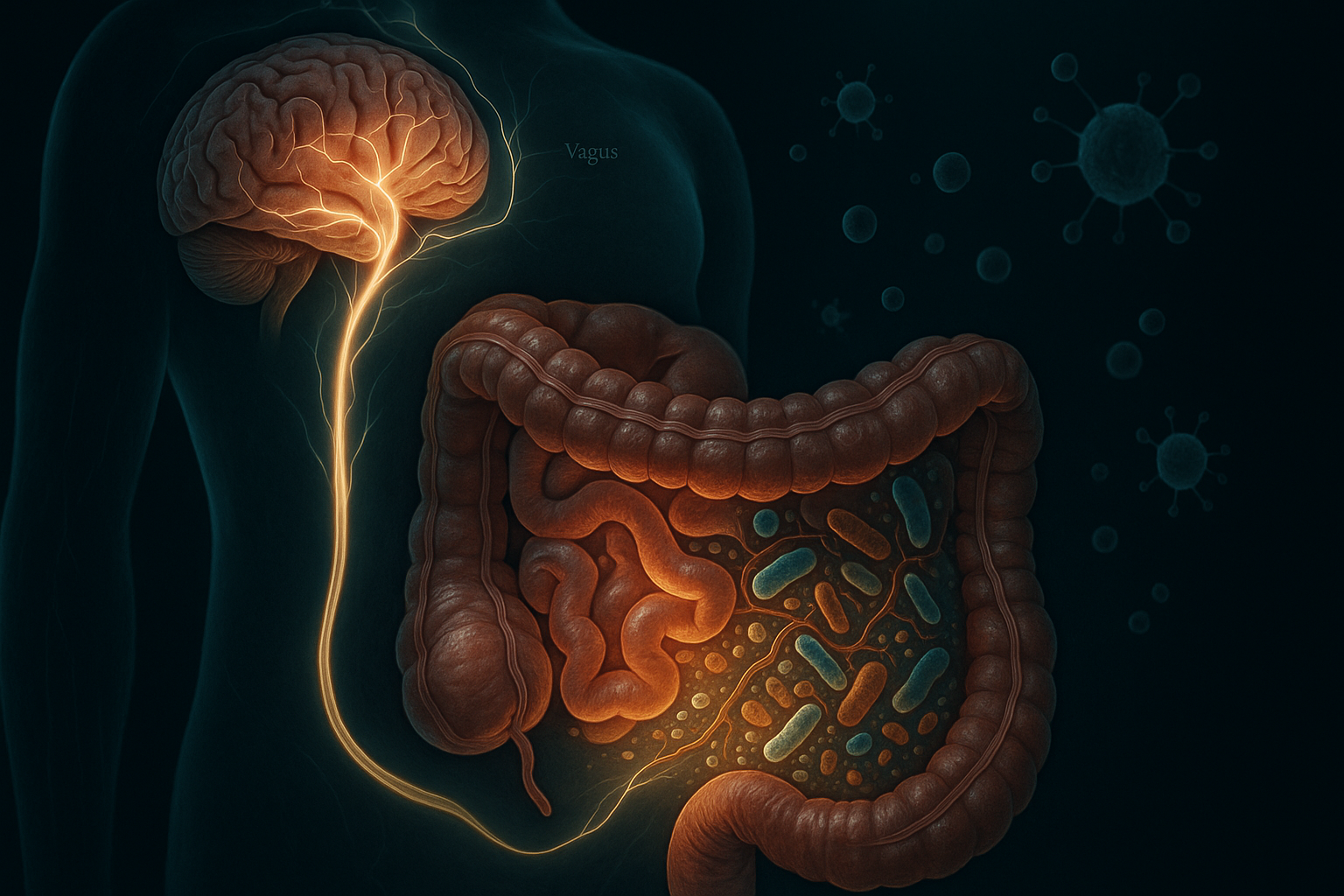
The gut-brain axis is a bidirectional communication network linking the intestinal microbiome to the central nervous system via the vagus nerve, enteric nervous system, hypothalamic-pituitary-adrenal (HPA) axis, and immune signalling pathways. Gut bacteria synthesise approximately 90% of the body's serotonin and influence dopamine, GABA, and cortisol regulation. Dysbiosis — reduced microbial diversity — is independently associated with generalised anxiety disorder and major depressive disorder in multiple human cohort studies, and emerging research in 2025 confirms gut microbiota dysfunction as a causative factor in mood disorders, not merely a consequence of them.
Your gut and your brain are in constant conversation. Every hour of every day, trillions of microbes living in your digestive tract send signals upward — influencing your mood, your stress response, your sleep, and your capacity to cope with the world. This is not wellness industry hype. It is one of the most rapidly advancing areas of modern neuroscience, with peer-reviewed research accumulating at pace.
This guide covers everything the science currently supports: the mechanisms of the gut-brain connection, what disrupts it, what restores it, and where to go deeper.
What's on this page
- What is the gut-brain axis?
- How your gut affects your mood
- The anxiety connection
- Foods that support gut and mental health
- Probiotics, prebiotics and postbiotics
- Signs your gut health may be affecting your mind
- What the science actually says
- Explore this topic in depth
What is the gut-brain axis?
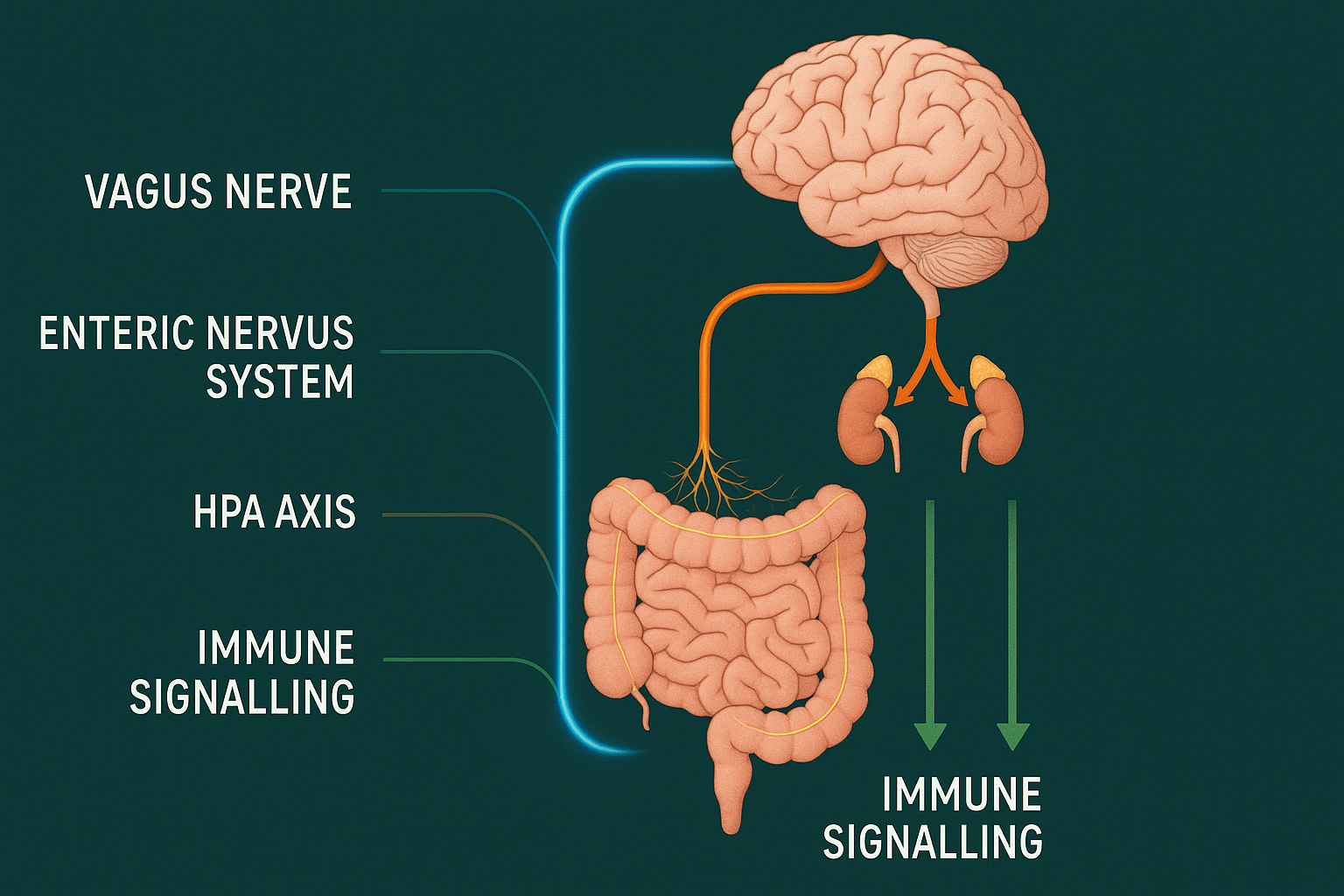
The gut-brain axis is a continuous two-way communication system linking the gastrointestinal tract to the brain. It is not a single pathway but a multi-channel network operating simultaneously through neural, hormonal, immune, and metabolic signals.
The primary channel is the vagus nerve — the longest cranial nerve in the body, running from the brainstem through the chest and abdomen to innervate the heart, lungs, and entire gastrointestinal tract. Approximately 80–90% of vagal fibres carry signals upward — from gut to brain — not the other way around. This asymmetry is important: it means the gut is, in neurological terms, primarily a sender. The vagus continuously monitors gut wall tension, mucosal chemistry, and microbial metabolites, transmitting that information to brain regions governing emotion, stress response, and decision-making.
But the vagus is only one channel. The gut-brain axis also communicates through:
- Neurotransmitters — serotonin, dopamine, GABA, and glutamate are all produced or influenced by gut bacteria
- Short-chain fatty acids (SCFAs) — compounds like butyrate and propionate produced when bacteria digest fibre, which cross the blood-brain barrier and exert direct neuroprotective effects
- The HPA axis — the hormonal stress-response system linking gut, adrenal glands, and brain; gut bacteria calibrate how sensitive this axis is to stress
- The immune system — gut bacteria regulate cytokine production that drives or suppresses neuroinflammation
Your gut also has its own autonomous nervous system — the enteric nervous system — with over 500 million neurons lining the digestive tract. It can operate independently of the brain and is sometimes called the "second brain." Its health and activity shape the quality of the signals reaching your brain continuously.
For a full scientific breakdown of how each pathway functions, see The Gut-Brain Axis: How Your Microbiome Controls Your Mood.
How your gut affects your mood
Here is what surprises most people: around 90–95% of the body's serotonin is produced in the gut, not the brain.
Serotonin is the neurotransmitter most associated with mood, emotional stability, and feelings of wellbeing. Gut-derived serotonin does not directly cross the blood-brain barrier — but it activates the vagus nerve, which carries those signals upward to influence emotional regulation and stress response. The bacteria responsible for driving serotonin synthesis include Clostridium species and Enterococcus faecalis, and their populations are directly shaped by diet, stress exposure, and sleep quality.
Gut bacteria are also directly involved in producing other key mood-regulating compounds:
- GABA — the brain's primary calming neurotransmitter, synthesised by Bifidobacterium and Lactobacillus species
- Dopamine precursors — gut bacteria including Bacillus species influence L-DOPA production and dopaminergic signalling
- Butyrate — a short-chain fatty acid that supports blood-brain barrier integrity, reduces neuroinflammation, and has been independently linked to improved mood and lower depression scores
- Tryptophan — gut bacteria regulate how much dietary tryptophan is converted to serotonin versus pro-inflammatory kynurenine pathway metabolites
When your microbiome is diverse and well-fed, this neurochemical production tends to work in your favour. When it is disrupted — by prolonged stress, a poor diet, antibiotic use, or illness — the downstream effects on mood can be significant and self-reinforcing. A 2025 study confirmed that gut microbiota dysbiosis functions as a causative factor in depression and anxiety, not merely a consequence of them — a distinction that matters enormously for how we understand and address mood disorders.
The anxiety connection
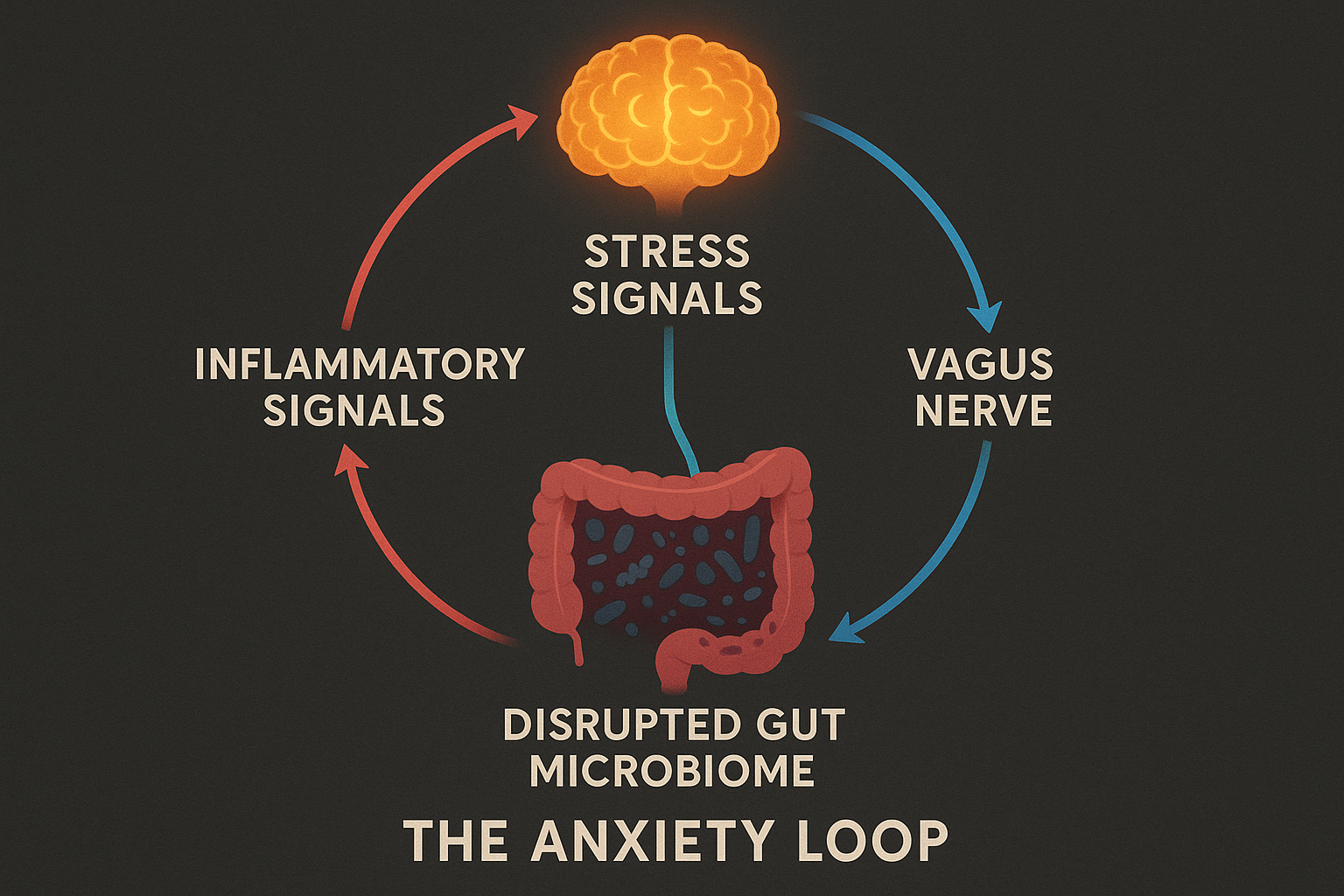
The gut-brain axis creates a feedback loop between stress and gut health that can become self-perpetuating. Stress signals from the brain alter gut motility, increase gut permeability, and shift the composition of your microbiome toward less beneficial bacteria. Those microbial changes then send distress signals back up to the brain via the vagus nerve, lowering the threshold for anxiety and making emotional regulation harder.
When the gut barrier becomes permeable — sometimes called "leaky gut" — bacterial endotoxins (lipopolysaccharides, or LPS) enter systemic circulation, triggering neuroinflammation. Elevated LPS is significantly associated with major depressive disorder in meta-analyses, and microglial activation driven by this pathway suppresses neurogenesis in the hippocampus — the brain region most associated with mood regulation and stress resilience.
Critically, this loop is interruptible. Diet, sleep quality, and targeted probiotic interventions are among the most studied means of doing so. See Gut Health and Anxiety: What the Science Really Says for the evidence-based interventions.
Foods that support gut and mental health
Diet is the single most powerful lever available for shaping the gut microbiome. What you eat determines which bacteria thrive, which decline, and what compounds they produce — with measurable changes occurring within 24 to 48 hours of a dietary shift.
The Mediterranean diet
The most researched dietary pattern for both gut health and mental wellbeing is the Mediterranean diet. Multiple randomised controlled trials have found it significantly reduces symptoms of depression and anxiety. The PREDIMED trial — one of the largest dietary intervention studies ever conducted — demonstrated sustained mental health benefits across eight years of follow-up. A 2025 meta-analysis confirmed that Mediterranean diet adherence produces statistically significant reductions in depressive symptoms in populations aged 18 to 65.
Its combination of high dietary fibre, polyphenol-rich plant foods, omega-3 fatty acids from oily fish, and fermented dairy makes it uniquely suited to supporting the gut-brain axis simultaneously through multiple pathways.
Foods with strong gut-brain evidence
Fermented foods — kefir, yogurt with live cultures, kimchi, sauerkraut, and kombucha deliver live bacteria that measurably increase microbiome diversity. A 2021 Stanford study found that a high-fermented-food diet increased microbiome diversity and reduced 19 inflammatory markers within 10 weeks.
Prebiotic-rich foods — garlic, onion, leeks, asparagus, oats, bananas, and Jerusalem artichokes feed beneficial bacteria already resident in the gut. Their fermentation by gut bacteria produces butyrate and other SCFAs that directly support brain health.
Oily fish — the omega-3 fatty acids EPA and DHA in salmon, sardines, and mackerel reduce neuroinflammation, support gut lining integrity, and have independent clinical evidence for reducing depression severity.
Dark chocolate (70%+ cocoa) — high in polyphenols that act as prebiotic fuel for beneficial bacteria; associated with reduced cortisol in acute stress studies.
Tryptophan-rich foods — turkey, eggs, salmon, seeds, and tofu supply tryptophan, the amino acid precursor to serotonin; the ratio converted to serotonin versus inflammatory metabolites is directly shaped by gut bacterial composition.
What undermines the gut-brain axis
Ultra-processed foods, refined sugars, and diets high in emulsifiers and trans fats consistently associate with reduced microbiome diversity, elevated inflammatory markers, and increased risk of mood disorders. Food emulsifiers including polysorbate 80 and carboxymethylcellulose directly damage the mucus layer protecting the gut epithelium — an effect demonstrated in both human and animal studies.
For specific foods and their clinical evidence ratings, see Best Foods for Gut Health and Mental Wellbeing.
Probiotics, prebiotics and postbiotics
These three terms are frequently conflated. They are distinct, and understanding the difference matters for making informed choices.
Probiotics are live microorganisms that confer a health benefit when consumed in adequate amounts. Not all probiotics are the same — strain specificity is critical. For mental health applications, the most studied strains belong to Lactobacillus and Bifidobacterium genera, some of which have demonstrated effects on anxiety and stress markers in human trials. These are sometimes called "psychobiotics." A 2023 meta-analysis in JAMA Psychiatry covering 34 randomised controlled trials found probiotic supplementation produced a statistically significant reduction in depression scores compared to placebo.
Prebiotics are dietary fibres that your body cannot digest but your gut bacteria can, serving as fermentation fuel. Prebiotic supplementation is building a strong evidence base for supporting mood independently of probiotics, with effects mediated largely through SCFA production and microbiome diversity increases.
Postbiotics are the bioactive compounds produced by bacteria when they ferment prebiotics — including butyrate, acetate, and propionate. They contain no live bacteria, making them more stable and potentially suitable where live cultures are contraindicated. Research on postbiotics as targeted mood interventions is among the most promising emerging areas in gut-brain science.
A food-first approach is generally recommended: meaningful prebiotic and probiotic support is achievable through diet alone for most people. Supplements are useful for specific therapeutic goals, but quality and strain specificity vary enormously across products. See Best Probiotics for Mental Health for evidence-ranked options.
Signs your gut health may be affecting your mind
These patterns do not constitute a clinical diagnosis. If you are concerned about mental health symptoms, please consult a GP or mental health professional. But these signals are worth recognising:
- Mood shifts that follow digestive events — feeling anxious or low after gut discomfort, or noticing digestive flare-ups during periods of psychological stress, reflects the bidirectional loop in operation
- Persistent low mood alongside IBS or bloating — co-occurrence of mood and gastrointestinal symptoms is common because both systems share the same dysregulated pathway
- Post-antibiotic mood changes — broad-spectrum antibiotics can reduce gut microbial diversity by up to 30% and alter gut-brain signalling for months; low mood or anxiety following a course is a recognised, if under-discussed, pattern
- Brain fog after eating — reduced mental clarity following meals, particularly high-sugar or ultra-processed meals, can reflect gut-brain signalling disruption and inflammatory response
- Declining stress resilience — feeling unable to recover from stressors that previously felt manageable; this can reflect HPA axis dysregulation driven by gut dysbiosis
- Sleep-mood co-fluctuation — nights of poor sleep that consistently precede worse mood the following day, and vice versa, reflect the gut-sleep-brain axis in disrupted function
For a detailed guide to identifying these patterns and understanding what they mean, see 7 Signs Your Gut Health Is Affecting Your Mental Health.

What the science actually says
This field is young and fast-moving. An honest summary of the current evidence:
Well-established by multiple large studies:
- The gut-brain axis exists and operates through four confirmed pathways: vagus nerve, enteric nervous system, HPA axis, and immune signalling
- Approximately 90–95% of serotonin is produced in the gut
- Gut microbiome composition is independently associated with anxiety and depression in human cohort studies
- Mediterranean diet reduces depression and anxiety symptoms in randomised controlled trials
- Gut dysbiosis precedes mood disorders in some individuals, functioning as a causative factor rather than a consequence
Promising but not yet definitive:
- Specific probiotic strains as clinical treatments equivalent to antidepressants — effect sizes in large trials are modest
- Personalised nutrition based on individual microbiome profiling — technically feasible but not yet clinically validated at scale
- Postbiotics as targeted mood interventions — strong mechanistic evidence, limited large-scale human trial data
Important caveats:
- Most microbiome-mood research has been in relatively small trials; effect sizes at population level are often modest
- The microbiome is highly individual — what produces measurable benefit for one person may not for another
- Gut health support is not a replacement for professional mental health care; it is a complementary strategy
Explore this topic in depth
Foundational guides
- The Gut-Brain Axis: How Your Microbiome Controls Your Mood — The complete neuroscience: vagus nerve, HPA axis, neuroinflammation, and all four communication pathways explained in full.
- Gut Health and Anxiety: What the Science Really Says — A deep dive into the bidirectional relationship between gut dysbiosis and anxiety disorders, with evidence-based interventions.
- Best Foods for Gut Health and Mental Wellbeing — Which foods have the strongest clinical evidence for improving the gut-brain axis, with evidence ratings for each.
Quick answers
- Can gut bacteria cause anxiety?
- How does the gut-brain axis work?
- What are the best probiotics for mental health?
- Can leaky gut cause depression?
- How long does it take to improve gut health for better mood?
From the blog
- 7 Signs Your Gut Health Is Affecting Your Mental Health — The overlooked physical and psychological signals that suggest your microbiome may be influencing your mood.
- Ozempic, Gut Health and Mental Wellbeing: What the Research Shows — How GLP-1 receptor agonists interact with the microbiome and what early data shows about mood effects.
- The Gut-Brain Diet: A 7-Day Eating Plan for Mental Wellbeing — A research-backed weekly meal framework designed to feed both your microbiome and your mind.
This page is for informational purposes only and does not constitute medical advice. If you are experiencing symptoms of anxiety, depression, or digestive conditions, please consult a qualified healthcare professional.
Sources
- Hwang YK, Oh JS. Interaction of the vagus nerve and serotonin in the gut-brain axis. Int J Mol Sci. 2025;26(3):1160. https://doi.org/10.3390/ijms26031160
- Lai Y, Xiong X. Gut microbiota dysbiosis as a causative factor in depression and anxiety. Front Psychiatry. 2025. PMC12585160.
- Sonnenburg JL et al. Diet-induced alterations in gut microflora contribute to lethal pulmonary damage in TLR2/TLR4-deficient mice. Cell. 2021. (High-fermented food trial reference.)
- Jacka FN et al. A randomised controlled trial of dietary improvement for adults with major depression (SMILES trial). BMC Medicine. 2017;15:23.
- Bizzozero-Peroni B et al. Mediterranean diet adherence and depressive symptoms: 2025 meta-analysis.
- PMC. Gut microbiota–immune–brain axis. Cell Rep Med. 2025;6(3):101982. https://pmc.ncbi.nlm.nih.gov/articles/PMC11970326/
- Thaiss CA, Levy M. Gut-brain connection, serotonin, and long COVID. Stanford Medicine. 2025. https://med.stanford.edu/news/insights/2025/03/gut-brain-connection-long-covid-anxiety-parkinsons.html
