How to Understand BRP Peptide for Weight Loss
Stanford's AI-discovered BRP peptide suppresses appetite like Ozempic but targets only the brain's hypothalamus — no nausea, no muscle loss, gut-friendly.
You've probably tried everything. Calorie counting, intermittent fasting, prescription appetite suppressants — and still, the weight creeps back. If you've considered Ozempic but paused because of the side effects — nausea, constipation, muscle wasting — you're not alone. Millions of people are caught between wanting effective help and dreading what comes with it.
Now, a naturally occurring molecule discovered by Stanford Medicine researchers is rewriting what appetite suppression can look like. It's called BRP, and it works through your brain's appetite-control centre — not your gut, pancreas, or digestive tract. That distinction matters enormously for how your body responds.
This guide walks you through exactly what BRP peptide is, why it works, how it differs from existing drugs, and what its discovery means for the gut-brain connection we now know drives so much of our hunger and metabolic health.
Why Appetite and Weight Management Are So Hard in the First Place
Appetite isn't just about willpower. It is a complex biological system orchestrated by hormones, neurons, and chemical signals that constantly shuttle information between your gut and your brain. When that system is dysregulated — by stress, poor diet, inflammation, or disrupted gut microbiome — hunger signals become louder than they should be.
- The gut-brain axis is the command centre. Hormones like GLP-1 (glucagon-like peptide 1) are produced in the gut and send satiety signals to the brain. When this communication breaks down, the brain fails to register fullness correctly.
- Existing drugs target broadly. Semaglutide (Ozempic) works by mimicking GLP-1, but GLP-1 receptors exist throughout the body — in the gut, pancreas, and other tissues — causing widespread effects including slowed digestion, nausea, and blood sugar swings.
- Gut microbiome disruption compounds the problem. Research increasingly shows that an imbalanced microbiome alters the production of appetite-regulating hormones, making weight management harder even before food passes your lips.
- The hypothalamus is the missing target. The brain region that truly controls long-term appetite and metabolism is the hypothalamus — and until BRP, no drug had found a clean, targeted path to it without collateral effects elsewhere.
Step 1: Understand What BRP Peptide Actually Is
BRP is a 12-amino-acid peptide — a tiny but extraordinarily potent protein fragment — discovered naturally in the human body. Its full name is BRINP2-related-peptide, derived from its parent protein BPM/retinoic acid inducible neural specific 2 (BRINP2).
According to research published in Nature in March 2025, BRP was identified using an AI-powered algorithm called Peptide Predictor, developed by Stanford Medicine researchers led by assistant professor of pathology Katrin Svensson, PhD. The algorithm screened all 20,000 human protein-coding genes and narrowed down a list of 373 prohormones — biologically inactive molecules that become active when cleaved into smaller peptide fragments.
From those, 100 peptides were screened for their ability to activate neuronal cells in the lab. GLP-1 tripled neuronal activity. BRP increased it tenfold. That extraordinary signal was the first clue that something remarkable had been found.
Why this matters for you: Unlike drugs that are engineered in a lab from scratch, BRP is something your body already makes. That suggests it may work with your physiology rather than forcing an artificial override — a critical distinction when thinking about long-term safety and tolerability.
Pro-tip: Understanding whether a molecule is endogenous (naturally occurring) versus synthetic is one of the most important questions to ask when evaluating any new treatment. Endogenous molecules often have better-tolerated profiles because the body already has mechanisms to process them.
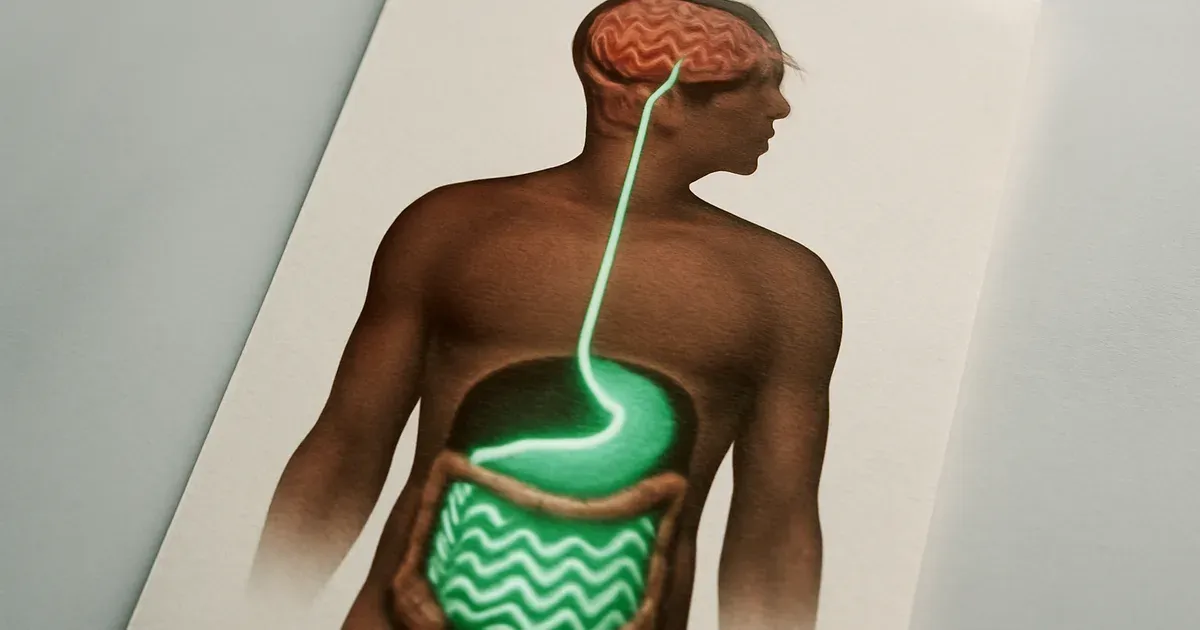
Step 2: Learn How BRP Targets the Gut-Brain Axis Differently
The gut-brain axis is a two-way motorway of chemical signals, nerve impulses, and hormonal messages connecting your digestive system to your brain. Most appetite-suppressing drugs, including semaglutide, intervene somewhere along this road — but often at multiple junctions simultaneously, causing traffic jams in the form of side effects.
BRP takes a different off-ramp. Rather than binding receptors in the gut, pancreas, or peripheral tissues, BRP appears to act specifically in the hypothalamus — the brain region that governs hunger, metabolism, and energy expenditure. This is a far more targeted destination on the gut-brain highway.
"The receptors targeted by semaglutide are found in the brain but also in the gut, pancreas and other tissues," said Svensson. "In contrast, BRP appears to act specifically in the hypothalamus, which controls appetite and metabolism."
This specificity has profound implications for your gut health. Because BRP does not slow the movement of food through the digestive tract — a key side effect of Ozempic — it leaves normal gut motility intact. This means your gut microbiome continues to receive food-derived signals on a normal schedule, avoiding the microbiome disruption that altered gut transit times can cause.
For anyone conscious of gut health, this is a meaningful difference. A healthy, diverse microbiome depends on consistent feeding rhythms, fibre fermentation, and uninterrupted gut motility. A drug that respects that rhythm is, in principle, friendlier to the ecosystem living inside you.
Step 3: Follow the Evidence From Animal Studies
The results in animals are striking. When BRP was injected into lean mice and minipigs — animals chosen because minipigs closely mirror human metabolism and eating patterns — food intake over the following hour dropped by up to 50% in both species.
Obese mice treated with daily BRP injections for 14 days lost an average of 3 grams, almost entirely from fat loss. Control animals gained approximately 3 grams over the same period. The treated mice also showed improved glucose and insulin tolerance — suggesting benefits beyond simple appetite suppression that extend to metabolic health more broadly.
Critically, Stanford Medicine researchers found no significant side effects in behavioural studies:
- No differences in movement or physical activity
- No changes in water intake
- No anxiety-like behaviour
- No changes in fecal production (confirming normal gut motility)
- No significant muscle mass loss
This last point deserves emphasis. One of the most concerning side effects of GLP-1 agonists like semaglutide is significant loss of lean muscle mass alongside fat. Muscle is metabolically active tissue — losing it slows your metabolism and makes weight regain more likely. BRP's fat-specific weight loss profile could make it a fundamentally different tool in the obesity treatment landscape.
Pro-tip: When evaluating any weight-loss intervention — drug, supplement, or dietary strategy — always ask whether the weight being lost is fat or muscle. The distinction has enormous long-term consequences for metabolic health.

Step 4: Understand What the Gut Microbiome Connection Means for Future Research
The gut microbiome produces and responds to appetite-regulating signals. Trillions of bacteria in your digestive tract communicate with your brain via the vagus nerve, immune signalling, and the production of short-chain fatty acids and neurotransmitters. When gut flora is imbalanced — a state called dysbiosis — this signalling goes awry, contributing to overeating and weight gain.
BRP's mechanism opens a fascinating research question: could an imbalanced gut microbiome be one reason BRP production or signalling is disrupted in people with obesity? Prohormone convertase 1/3 — the enzyme that cleaves BRP from its parent protein — is already known to be involved in human obesity. It is plausible that gut-derived inflammatory signals from a disrupted microbiome interfere with prohormone processing, reducing natural BRP availability.
Researchers are now working to identify the cell-surface receptors that bind BRP and to extend the peptide's effective duration in the body for more practical dosing. They are also investigating its full pathway of action — research that may ultimately reveal precisely how gut-brain communication shapes BRP's role in energy regulation.
For the gut health community, this is an invitation to think about appetite not as a failure of willpower but as a biological conversation — one that involves your microbiome, your gut hormones, your vagus nerve, and your hypothalamus. Supporting that conversation through diet, fibre, fermented foods, and reduced gut inflammation remains one of the most powerful things you can do right now, while BRP moves toward human trials.
Timeline: What to Expect as BRP Moves Toward Humans
Phase 1 — Animal Validation (Completed, 2025) Stanford researchers have confirmed appetite suppression, fat-specific weight loss, improved glucose and insulin tolerance, and absence of major side effects in mice and minipigs.
Phase 2 — Receptor Identification (Ongoing) Scientists are mapping the exact cell-surface receptors BRP binds to, which will clarify mechanism and guide dosing. This step is critical for designing safe human trials.
Phase 3 — Extended Duration Research (Ongoing) Peptides are naturally short-lived in the body. Researchers are engineering modifications to help BRP last longer — essential for a practical treatment schedule.
Phase 4 — Human Clinical Trials (Near Future) Svensson has co-founded Merrifield Therapeutics to advance BRP toward clinical trials. Human safety and efficacy data will be the decisive test of whether BRP's animal-study promise translates to people.

Mistakes That Slow Your Progress While Waiting for New Science
- Ignoring your gut microbiome now. The gut-brain axis is active today, not just in clinical trial results. A fibre-poor, ultra-processed diet degrades the microbial diversity that supports healthy appetite signalling.
- Treating weight loss as a willpower problem. Hunger is hormonal. Blaming yourself for appetite dysregulation caused by biological factors keeps you stuck and demoralised.
- Expecting one drug to do everything. Even if BRP proves effective in humans, it will likely work best alongside lifestyle strategies that support gut-brain health — not instead of them.
- Dismissing early-stage science. Some readers will wait until BRP has a pharmacy shelf before taking it seriously. But understanding the mechanism now helps you make smarter decisions about your health today.
- Overlooking muscle loss as a weight-loss metric. If you are currently using or considering any appetite suppressant, track body composition, not just the number on the scale. Preserving muscle while losing fat is the true goal.
What Can Help You Get There Faster
Gut-Brain Nutrition Tools A diet rich in prebiotic fibre (oats, leeks, garlic, Jerusalem artichokes) feeds the bacteria that produce short-chain fatty acids — molecules that themselves support satiety signalling. Fermented foods like kefir, kimchi, and yoghurt support microbial diversity linked to healthier appetite regulation.
Lifestyle Supports for Appetite Hormones Sleep deprivation elevates ghrelin (the hunger hormone) and suppresses leptin (the satiety hormone). Prioritising 7–9 hours of sleep is one of the most evidence-supported ways to support your body's own appetite-regulating peptide system — including the prohormone convertase pathways that produce molecules like BRP.
Tracking and Awareness Resources Continuous glucose monitors, gut microbiome testing kits, and nutrition tracking apps can give you real-time feedback on how your dietary choices are influencing the gut-brain signals that drive hunger. Knowledge of your own patterns is the first step to changing them.
Summary: Your BRP Peptide Guide at a Glance
- ✅ Step 1: Understand that BRP is a naturally occurring 12-amino-acid peptide, discovered using AI, that activates brain neurons at 10× the potency of GLP-1
- ✅ Step 2: Recognise that BRP targets the hypothalamus specifically, leaving gut motility and the gut microbiome undisturbed — unlike semaglutide
- ✅ Step 3: Review the animal evidence: up to 50% food intake reduction, fat-specific weight loss, improved glucose tolerance, and no significant side effects
- ✅ Step 4: Connect BRP's mechanism to the broader gut-brain axis and support that system through nutrition, sleep, and microbiome care right now
- ✅ Timeline awareness: Human clinical trials are approaching — stay informed via Merrifield Therapeutics and ongoing peer-reviewed research
- ✅ Avoid: Blaming willpower, neglecting gut health, ignoring muscle loss, and dismissing promising early-stage science
Frequently Asked Questions
What is BRP peptide and how was it discovered?
BRP (BRINP2-related-peptide) is a naturally occurring 12-amino-acid peptide identified by Stanford Medicine researchers using an AI algorithm called Peptide Predictor. The algorithm screened all 20,000 human protein-coding genes to find peptides cleaved by prohormone convertase 1/3, narrowing 373 candidate prohormones down to BRP as the most potent activator of appetite-regulating neurons.
How is BRP different from Ozempic (semaglutide)?
Semaglutide mimics GLP-1 and binds receptors throughout the body — including the gut and pancreas — causing nausea, slowed digestion, and muscle loss. BRP appears to act specifically in the hypothalamus, the brain's appetite-control centre, activating separate neuronal pathways without the widespread peripheral effects that cause Ozempic's side effects.
Does BRP affect the gut microbiome?
Because BRP does not slow gut motility — unlike semaglutide — it leaves normal digestive rhythms intact. This is significant for gut microbiome health, since consistent gut transit times support microbial diversity and the production of appetite-regulating short-chain fatty acids. However, direct microbiome studies of BRP have not yet been published.
When will BRP be available for humans?
BRP is not yet approved for human use. Katrin Svensson has co-founded Merrifield Therapeutics to advance BRP toward clinical trials in the near future. Researchers are currently working on identifying its receptors and extending its duration of action in the body before human trials begin.
Can I support my appetite-regulating hormones naturally while waiting?
Yes. The same prohormone convertase pathways that produce BRP are influenced by your overall metabolic health. Eating a high-fibre diet to support gut microbiome diversity, prioritising sleep to regulate ghrelin and leptin, and reducing ultra-processed food intake all support the gut-brain signalling system that BRP targets — giving you meaningful tools to use right now.
