Gut Health & Immunity: Your Top Questions Answered
Discover how gut health, the microbiome, and the gut-brain axis shape immunity and infection risk — with science-backed answers to your top questions.
The connection between your gut and your immune system can feel overwhelming to understand. You've probably heard that gut health matters, but why exactly does it affect whether you get sick — and how? This article cuts through the noise, answering the biggest questions about the gut microbiome, mucosal immunity, and what science says about protecting yourself through nutrition. Whether you're curious about the gut-brain axis or how bacteria in your intestines fight infections, you'll find clear, evidence-based answers here.
Jump to Your Question
What is the gut microbiome and why does it matter for immunity?
How does the gut protect the body from infections?
What is colonization resistance and how does it work?
How does gut health affect systemic immunity beyond the intestines?
What is the gut-brain axis and does it influence immune responses?
Do antibiotics damage the gut microbiome and raise infection risk?
How does nutrition shape the gut microbiome and immune defenses?
Are infants and the elderly more vulnerable because of gut health?
What is the gut microbiome and why does it matter for immunity?
The gut microbiome is a vast community of bacteria, fungi, and viruses living inside the intestines that directly shapes how the immune system develops and responds. Roughly 70–80% of all immune cells in the human body are found in the gut, making the intestinal environment the largest immune organ in the body.
Gut microbes and humans have co-evolved over millions of years. Microbes receive a warm, nutrient-rich habitat, while in return they regulate critical host functions — including protective immunity against pathogens. This is not a passive relationship; the microbiome actively communicates with immune cells through molecular signals.
The composition of the gut microbiome is shaped by:
- Genetics and age
- Diet and nutrition
- Stress levels
- Antibiotic and drug use
- Environmental exposures (the "exposome")
When this community is balanced, it supports immune readiness. When it is disrupted, vulnerability to infection rises significantly.
How does the gut protect the body from infections?
The gut uses three interconnected lines of defense to block pathogens: the microbiota itself, the epithelial barrier, and the mucosal immune system. A pathogen must overcome all three to establish a true infection in the gastrointestinal tract.
The intestinal epithelium is a single-cell-thick layer joined by tight-junction proteins, forming a physical wall between gut contents and underlying tissues. This barrier is reinforced by a mucus layer that prevents bacteria from directly contacting epithelial cells.
Mucus does more than just block entry. It also acts as a reservoir for antimicrobial molecules such as secretory IgA (an antibody found in mucosal surfaces) and defensins — proteins that punch holes in bacterial membranes. Both the microbiota and the host inflammatory state influence how much mucus is produced and how effective it is.
Disruption of any one of these layers — through toxins released by harmful bacteria, chronic stress, or poor diet — can increase intestinal permeability and raise susceptibility to infection.

What is colonization resistance and how does it work?
Colonization resistance is the ability of the resident gut microbiota to prevent invading pathogens from establishing themselves in the intestine. It works primarily through competition: commensal bacteria and harmful microorganisms compete for the same nutrients and physical niches.
One key mechanism within colonization resistance is quorum sensing — a bacterial communication system in which microbes release chemical signals that accumulate as population density grows. When signal concentrations reach a threshold, bacteria collectively change their behavior, adjusting gene expression related to motility, adherence, and the secretion of protective compounds.
Commensals use quorum sensing to maintain gut homeostasis. Pathogens, however, exploit the same system to suppress host immune responses and enhance their own virulence. This creates an ongoing molecular negotiation happening continuously in the intestinal environment.
| Defense Mechanism | Primary Function | Disrupted By |
|---|---|---|
| Colonization resistance | Blocks pathogen colonization | Antibiotics, poor diet |
| Tight junction integrity | Physical barrier | Bacterial toxins, stress |
| Mucus layer | Traps pathogens | Dysbiosis, inflammation |
| Secretory IgA | Neutralizes antigens | Malnutrition, immune deficiency |
How does gut health affect systemic immunity beyond the intestines?
Gut health influences not just local mucosal defenses but also the systemic immune system — meaning the immune responses that protect every organ and tissue in the body. It is now well-recognized that the gut microbiome communicates with immune cells throughout the bloodstream and lymphatic system.
The gut contains both innate and adaptive immune components. The innate immune system provides rapid, nonspecific defenses through macrophages, natural killer cells, and physical barriers. The adaptive immune system — composed of T and B lymphocytes — mounts targeted responses to specific pathogens.
T cells recognize infected host cells and coordinate responses, while B cells produce antibodies that circulate through body fluids (humoral immunity). The gut microbiome influences both arms:
- Microbial metabolites (such as short-chain fatty acids) travel through the bloodstream and modulate immune cell activity in distant tissues.
- Dysbiosis (an imbalanced microbiome) is associated with low-grade systemic inflammation, reducing the precision and speed of immune responses.
- Leaky gut (increased intestinal permeability) allows microbial fragments to enter the bloodstream, triggering chronic immune activation.
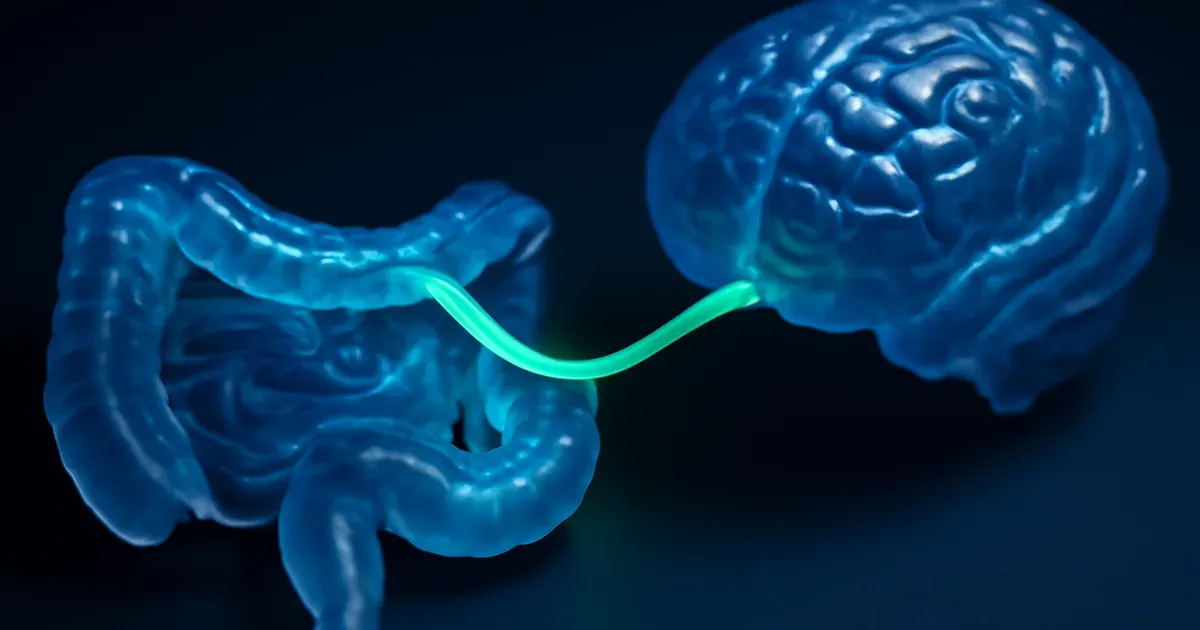
What is the gut-brain axis and does it influence immune responses?
The gut-brain axis is a bidirectional communication network linking the central nervous system with the enteric nervous system of the gut, and the microbiome sits at the center of this connection. Far from being a one-way relationship, the brain influences gut function and the gut feeds information back to the brain — with immune consequences at every step.
Stress signals from the brain can alter gut motility, mucus production, and the composition of the microbiome itself, all of which affect infection susceptibility. Chronic psychological stress has been shown to reduce microbial diversity and weaken the gut epithelial barrier.
In the other direction, gut microbes produce neurotransmitters (including serotonin, with around 90% of the body's serotonin made in the gut) and short-chain fatty acids that signal through the vagus nerve to the brain. This gut-to-brain signaling influences mood, stress regulation, and — critically for immunity — the inflammatory tone of the entire body.
The practical implication: managing stress is not just a mental health strategy. It is a genuine gut health and immune defense strategy, because the gut-brain axis directly links emotional states to intestinal barrier integrity and microbial balance.
Do antibiotics damage the gut microbiome and raise infection risk?
Antibiotics reduce infection risk from their target pathogen but simultaneously disrupt the gut microbiome, potentially raising vulnerability to other infections. By killing broad populations of bacteria — including beneficial commensals — antibiotics create ecological gaps that opportunistic pathogens can exploit.
This disruption directly reduces colonization resistance. Without a dense, competitive community of commensal microbes, harmful organisms like Clostridioides difficile can colonize, overgrow, and cause serious illness. This is one of the most well-documented consequences of antibiotic-associated gut dysbiosis.
Key effects of antibiotics on gut immunity include:
- Reduced microbial diversity, which may persist for months
- Impaired quorum sensing among commensals
- Decreased short-chain fatty acid production, weakening mucosal defenses
- Altered immune cell training in the gut-associated lymphoid tissue
Recovery of the microbiome after antibiotics varies by individual and antibiotic type. Dietary strategies — particularly increasing fiber and fermented food intake — are among the most evidence-supported approaches to supporting microbiome restoration.
How does nutrition shape the gut microbiome and immune defenses?
Nutrition is one of the most powerful modifiable factors influencing gut microbiome composition and, by extension, immune function. What you eat determines which microbial species thrive in the gut — and those species determine how well your immune defenses perform.
Dietary fiber, for example, serves as a prebiotic — a substrate fermented by gut bacteria to produce short-chain fatty acids like butyrate. Butyrate directly fuels intestinal epithelial cells, strengthens tight junctions, and has anti-inflammatory effects on immune cells both locally and systemically.
Diets that are:
- High in fiber and plant diversity → support microbial diversity and mucosal immunity
- High in ultra-processed foods and sugar → reduce beneficial bacteria and increase low-grade gut inflammation
- Rich in fermented foods (yogurt, kefir, kimchi) → may directly introduce beneficial microbes and support existing communities
Micronutrients also play a direct role. Vitamins A, C, D, and zinc are all essential for maintaining epithelial barrier integrity, mucus production, and immune cell function. Deficiencies in these nutrients — common in low-income settings — help explain why infectious disease burden remains highest in those populations.
Are infants and the elderly more vulnerable because of gut health?
Infants and elderly individuals face higher infection risk partly because their gut microbiomes and associated immune defenses are at different stages of development or decline. Infectious diarrhea, respiratory infections, influenza, and pneumonia disproportionately affect both groups worldwide.
In infants, the gut microbiome is still being established. Colonization by the first microbes — influenced by birth mode, breastfeeding, and early environment — shapes immune system development for years. Disruption during this critical window (through early antibiotic use, formula feeding, or poor diet) can impair immune training and increase susceptibility to enteric and respiratory infections.
In the elderly, microbiome diversity tends to decline with age, alongside reductions in tight junction protein expression, mucus quality, and the responsiveness of immune cells. This combination of gut barrier weakening and immune senescence explains why influenza and pneumonia remain common causes of death in older populations even in high-income countries.
Supporting gut health across the lifespan — through diet, mindful antibiotic use, and stress management — represents one of the most accessible strategies for reducing infection risk in both vulnerable groups.
The Bottom Line
- 70–80% of immune cells live in the gut, making gut health inseparable from immune defense.
- Three barriers protect against infection: the microbiota, the epithelial layer, and the mucosal immune system — all of which depend on a healthy microbiome.
- The gut-brain axis links stress, mood, and gut barrier integrity, meaning psychological wellbeing has direct immune consequences.
- Antibiotics disrupt colonization resistance, creating windows of vulnerability to opportunistic pathogens.
- Diet is the most accessible lever: fiber, fermented foods, and key micronutrients all support the microbiome and immune readiness across every life stage.
